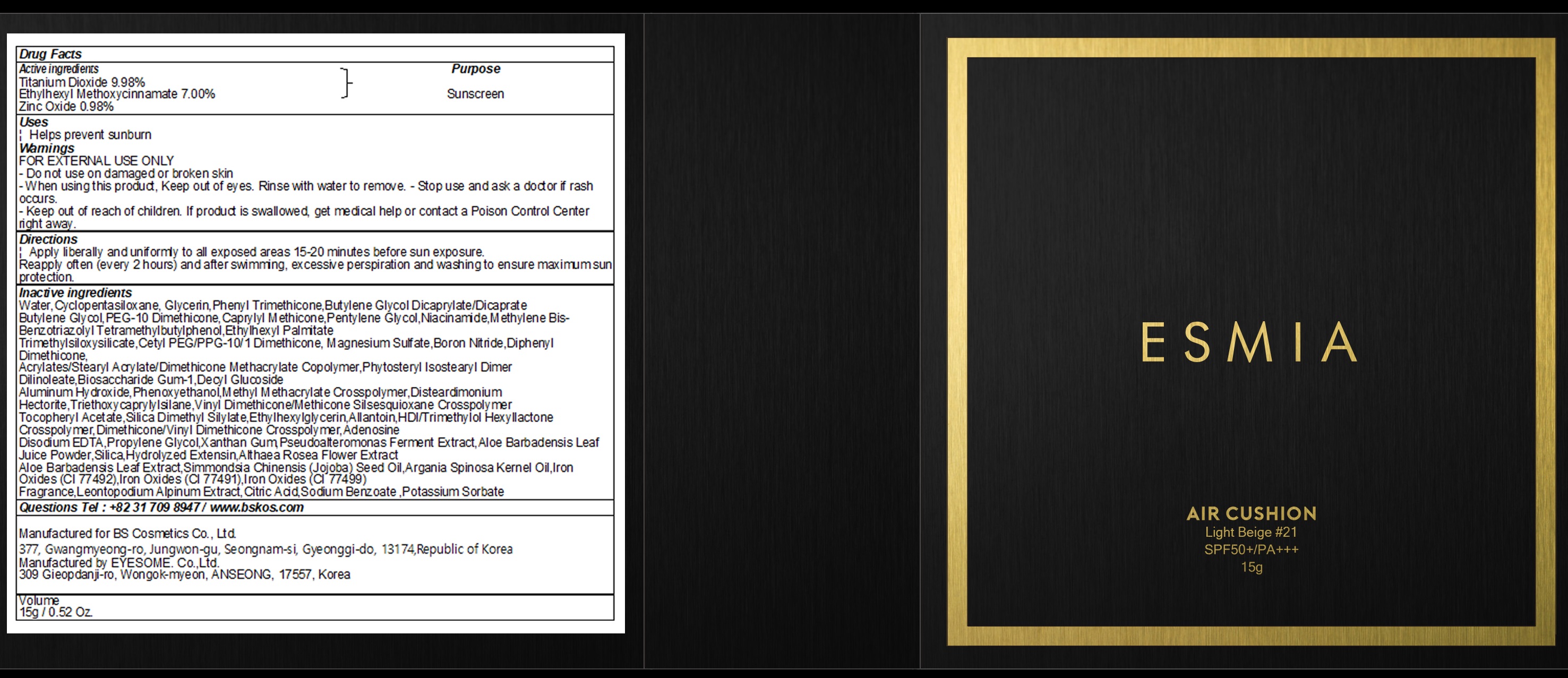
Label: ESMIA AIR CUSHION LIGHT BEIGE 21- titanium dioxide, octinoxate, zinc oxide powder
-
Contains inactivated NDC Code(s)
NDC Code(s): 72600-190-01, 72600-190-02 - Packager: Bs Cosmetics Co.,ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 19, 2018
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
-
INACTIVE INGREDIENT
Inactive ingredients:
Water,Cyclopentasiloxane, Glycerin,Phenyl Trimethicone,Butylene Glycol Dicaprylate/Dicaprate
Butylene Glycol,PEG-10 Dimethicone,Caprylyl Methicone,Pentylene Glycol,Niacinamide,Methylene Bis-Benzotriazolyl Tetramethylbutylphenol,Ethylhexyl Palmitate
Trimethylsiloxysilicate,Cetyl PEG/PPG-10/1 Dimethicone, Magnesium Sulfate,Boron Nitride,Diphenyl Dimethicone,
Acrylates/Stearyl Acrylate/Dimethicone Methacrylate Copolymer,Phytosteryl Isostearyl Dimer Dilinoleate,Biosaccharide Gum-1,Decyl Glucoside
Aluminum Hydroxide,Phenoxyethanol,Methyl Methacrylate Crosspolymer,Disteardimonium Hectorite,Triethoxycaprylylsilane,Vinyl Dimethicone/Methicone Silsesquioxane Crosspolymer
Tocopheryl Acetate,Silica Dimethyl Silylate,Ethylhexylglycerin,Allantoin,HDI/Trimethylol Hexyllactone Crosspolymer,Dimethicone/Vinyl Dimethicone Crosspolymer,Adenosine
Disodium EDTA,Propylene Glycol,Xanthan Gum,Pseudoalteromonas Ferment Extract,Aloe Barbadensis Leaf Juice Powder,Silica,Hydrolyzed Extensin,Althaea Rosea Flower Extract
Aloe Barbadensis Leaf Extract,Simmondsia Chinensis (Jojoba) Seed Oil,Argania Spinosa Kernel Oil,Iron Oxides (CI 77492),Iron Oxides (CI 77491),Iron Oxides (CI 77499)
Fragrance,Leontopodium Alpinum Extract,Citric Acid,Sodium Benzoate ,Potassium Sorbate - Purpose
-
Warnings
Warnings:
FOR EXTERNAL USE ONLY
- Do not use on damaged or broken skin
- When using this product, Keep out of eyes. Rinse with water to remove.
- Stop use and ask a doctor if rash occurs.
- Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
- DESCRIPTION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ESMIA AIR CUSHION LIGHT BEIGE 21
titanium dioxide, octinoxate, zinc oxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72600-190 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 1.49 g in 15 g Octinoxate (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) Octinoxate 1.05 g in 15 g Zinc Oxide (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 0.80 g in 15 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72600-190-02 1 in 1 CARTON 09/01/2018 1 NDC:72600-190-01 15 g in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 09/01/2018 Labeler - Bs Cosmetics Co.,ltd. (694791429) Registrant - Bs Cosmetics Co.,ltd. (694791429) Establishment Name Address ID/FEI Business Operations EYESOME. Co.,Ltd. 557795360 manufacture(72600-190)