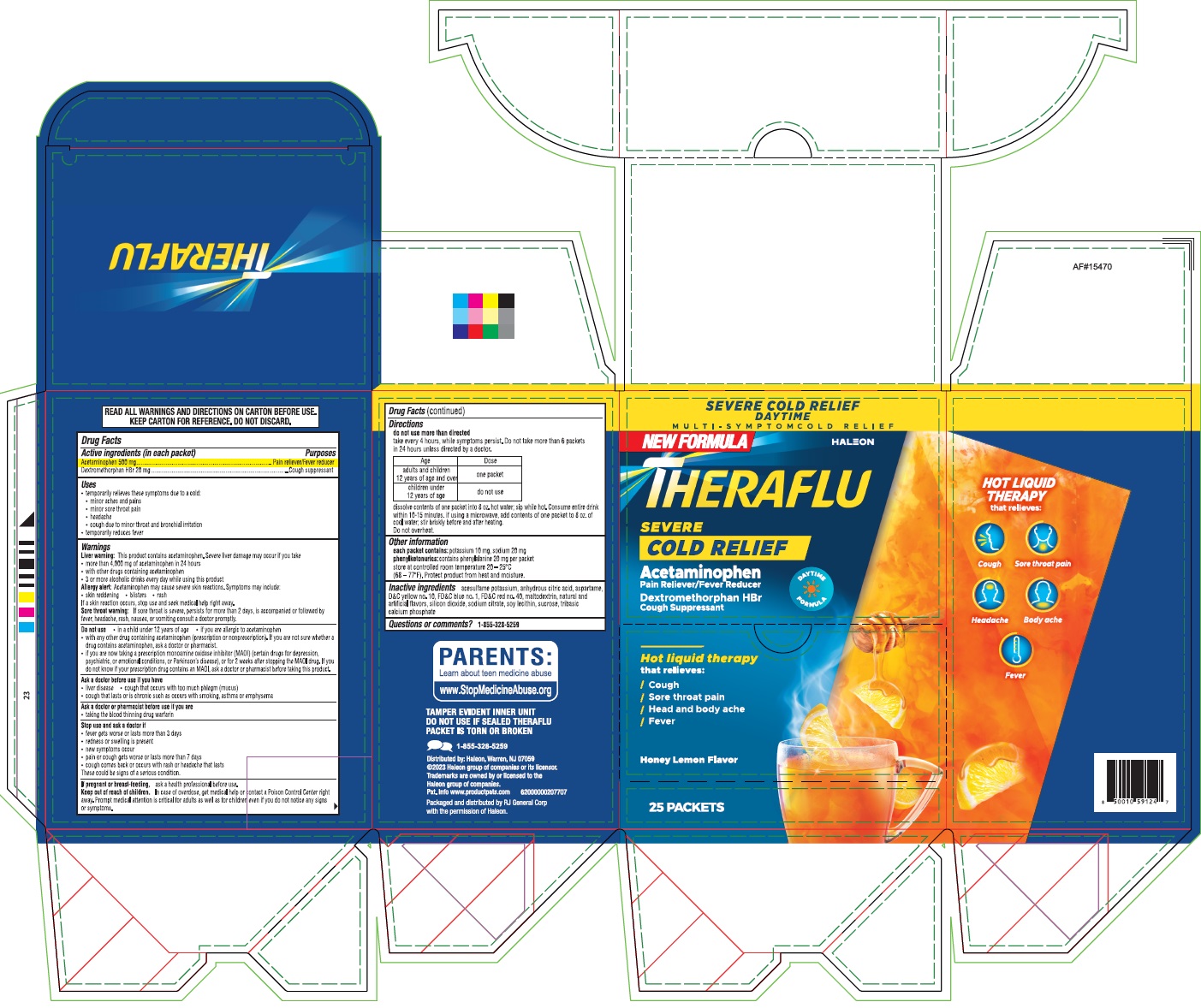
Label: THERAFLU SEVERE COLD RELIEF DAYTIME- acetaminophen, dextromethorphan hbr powder, for solution
- NDC Code(s): 70264-047-01
- Packager: R J General Corporation
- This is a repackaged label.
- Source NDC Code(s): 0067-0100
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 15, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each packet)
- PURPOSE
- Uses
-
Warnings
Liver warning:This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert:Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning:If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting consult a doctor promptly.
Do not use
- in a child under 12 years of age
- if you are allergic to acetaminophen
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or a pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask doctor before use if you have
- liver disease
- cough that occurs with too much phlegm (mucus)
- cough that lasts or is chronic such as occurs with smoking, asthma or emphysema
-
Directions
- do not use more than directed
- take every 4 hours, while symptoms persist. Do not take more than 6 packets in 24 hours unless directed by a doctor.
Age
Dose
adults and children
12 years of age and over
one packet
children under
12 years of age
do not use
- dissolve contents of one packet into 8 oz. hot water; sip while hot. Consume entire drink within 10-15 minutes. if using a microwave, add contents of one packet to 8 oz. of cool water; stir briskly before and after heating.
- Do not overheat.
- Other information
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
THERAFLU SEVERE COLD RELIEF DAYTIME
acetaminophen, dextromethorphan hbr powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70264-047(NDC:0067-0100) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ASPARTAME (UNII: Z0H242BBR1) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) MALTODEXTRIN (UNII: 7CVR7L4A2D) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) SUCROSE (UNII: C151H8M554) TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) Product Characteristics Color white (to off white, yellow and brown) Score Shape Size Flavor HONEY (Lemon) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70264-047-01 25 in 1 CARTON 11/12/2024 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 11/12/2024 Labeler - R J General Corporation (122542830) Establishment Name Address ID/FEI Business Operations R J General Corporation 122542830 repack(70264-047)