Label: PREMIER VALUE ZINC COLD THERAPY CITRUS FLAVOR- zinc gluconate tablet
- NDC Code(s): 68016-118-25
- Packager: PHARMACY VALUE ALLIANCE, LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 3, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
Uses
- ▪
- may help reduce duration and severity of cold symptoms:
- ▪
- sore throat
- ▪
- stuffy nose
- ▪
- sneezing
- ▪
- coughing
- This product was formulated to help reduce the duration and severity of cold symptoms and was not formulated to be effective for flu or allergies.
-
Directions
- ▪
- for best results, use at the first sign of a cold and continue to use as directed
- ▪
- adults and children 12 years of age and older:
- ▪
- chew one tablet completely at the onset of symptoms. Do not swallow whole.
- ▪
- repeat every 3 hours until symptoms are gone
- ▪
- to avoid minor stomach upset, do not take on an empty stomach.
- ▪
- do not eat or drink for 15 minutes after use. Do not eat or drink citrus fruits or juices for 30 minutes before or after use. Otherwise, drink plenty of fluids.
- ▪
- recommended daily dose is 6 tablets for adults and 4 tablets for ages 12 through 17
- ▪
- Children under 12 years of age: Consult a doctor before use.
- Other information
- Inactive ingredients
- Questions or comments?
-
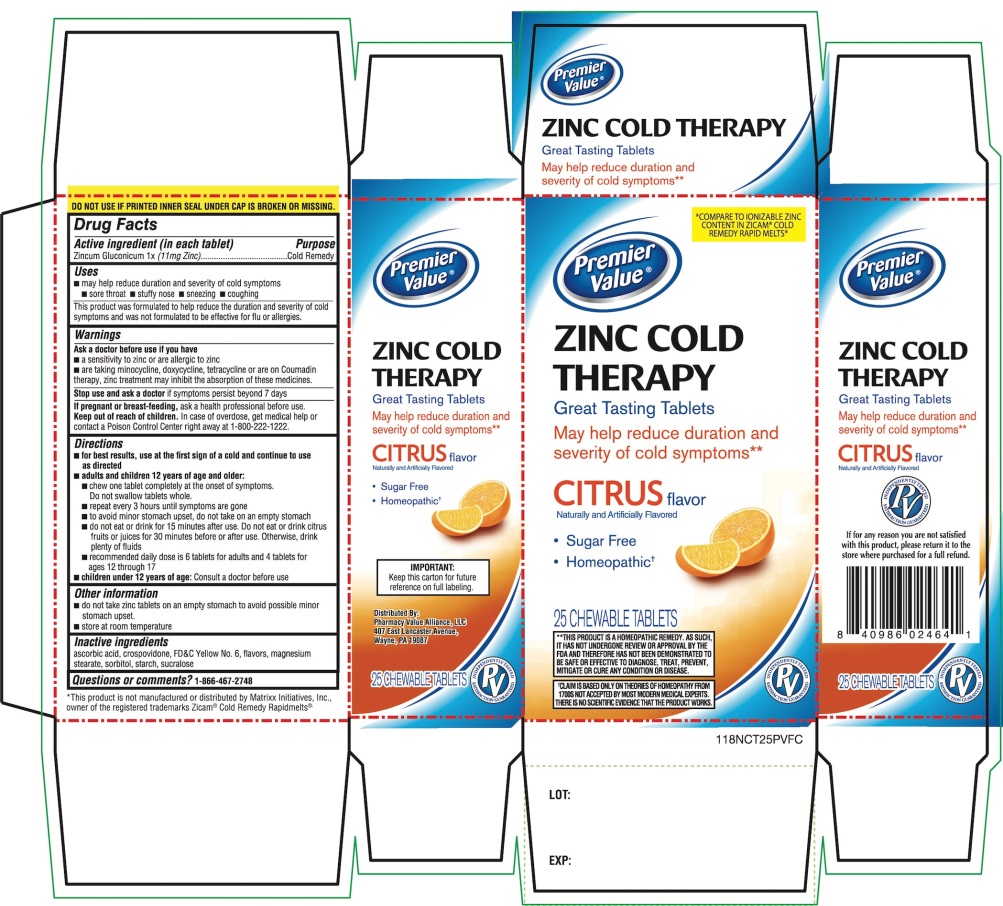
PRINCIPAL DISPLAY PANEL
Premier Value®
NDC 68016-118-25
*COMPARE TO ® IONIZABLE ZINC CONTENT IN ZICAM® COLD REMEDY RAPID MELTS*
ZINC COLD THERAPY
Great Tasting TabletsMay help reduce the duration and severity of cold symptoms**
Citrus Flavor
Naturally and Artificially Flavored
- •
- Sugar Free
- •
- Homeopathic†
25 CHEWABLE TABLETS
DO NOT USE IF PRINTED INNER SEAL UNDER CAP IS BROKEN OR MISSING.
†CLAIM IS BASED ONLY ON THEORIES OF HOMEOPATHY FROM 1700S NOT ACCEPTED BY MOST MODERN MEDICAL EXPERTS .THERE IS NO SCIENTIFIC EVIDENCE THAT THE PRODUCT WORKS
**THIS PRODUCT IS A HOMEOPATHIC REMEDY, AS SUCH, IT HAS NOT UNDERGONE REVIEW ON APPROVAL BY THE FDA AND THEREFORE HAS NOT BEEN DEMONSTRATED TO BE SAFE OR EFFECTIVE TO DIAGNOSE, TREAT, PREVENT, MITIGATE OR CURE ANY CONDITION OR DISEASE.
IMPORTANT: Keep this carton for future reference on full labeling.
*This product is not manufactured or distributed by Matrixx Initiatives, Inc., owner of the registered trademarks Zicam® Cold Remedy Rapidmelts®
Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
-
INGREDIENTS AND APPEARANCE
PREMIER VALUE ZINC COLD THERAPY CITRUS FLAVOR
zinc gluconate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-118 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC GLUCONATE (UNII: U6WSN5SQ1Z) (ZINC CATION - UNII:13S1S8SF37) ZINC GLUCONATE 1 [hp_X] Inactive Ingredients Ingredient Name Strength ascorbic acid (UNII: PQ6CK8PD0R) CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) FD&C Yellow No. 6 (UNII: H77VEI93A8) magnesium stearate (UNII: 70097M6I30) SORBITOL (UNII: 506T60A25R) STARCH, CORN (UNII: O8232NY3SJ) sucralose (UNII: 96K6UQ3ZD4) Product Characteristics Color ORANGE Score no score Shape ROUND (Biconcave) Size 16mm Flavor CITRUS Imprint Code RP118 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-118-25 25 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/18/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved homeopathic 04/18/2019 Labeler - PHARMACY VALUE ALLIANCE, LLC (101668460)