Label: LEADER EARWAX REMOVAL KIT- carbamide peroxide 6.50% kit liquid
- NDC Code(s): 70000-0490-1, 70000-0490-2
- Packager: Cardinal Health, 110 dba Leader
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 22, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
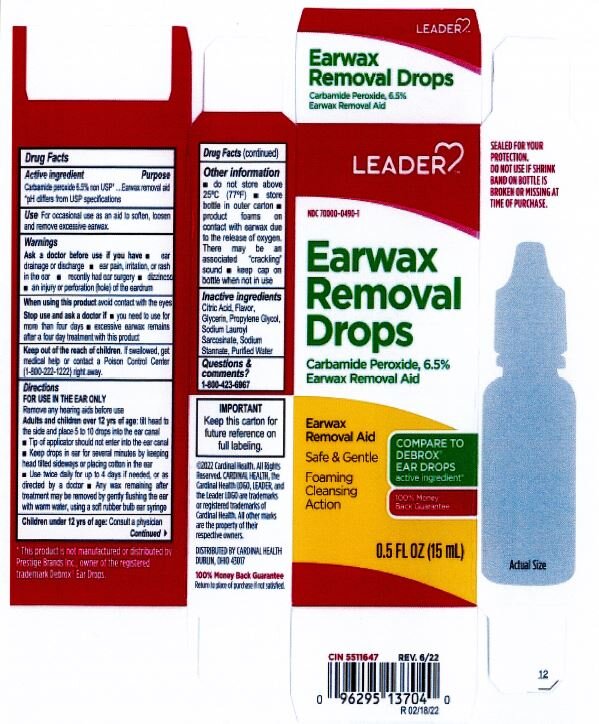
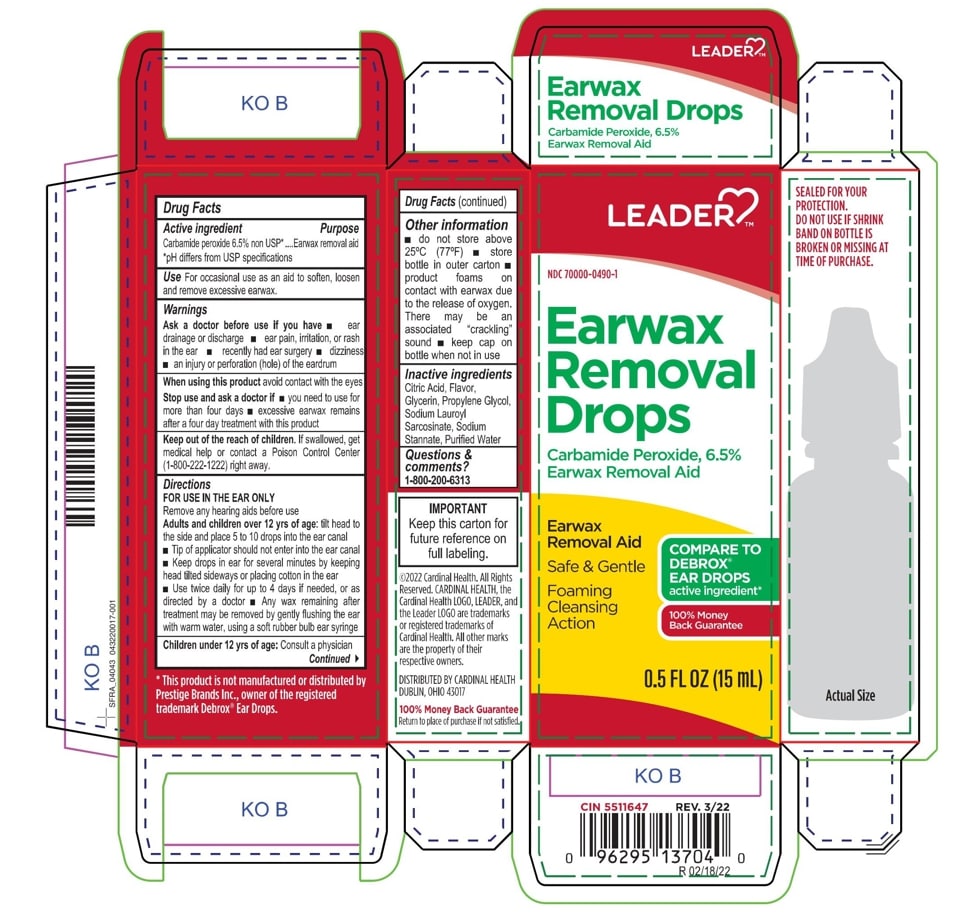
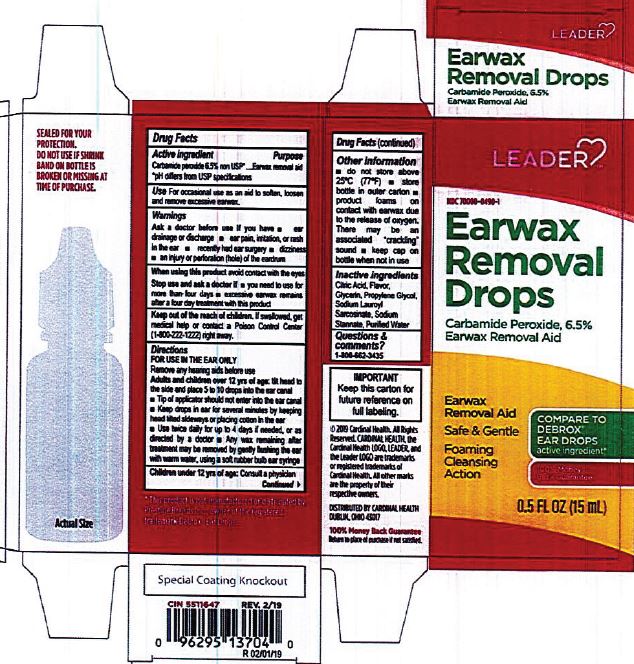
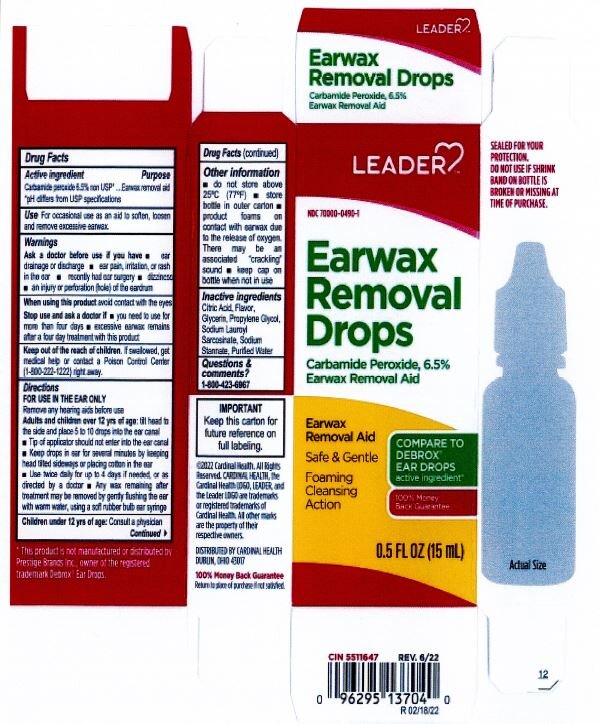
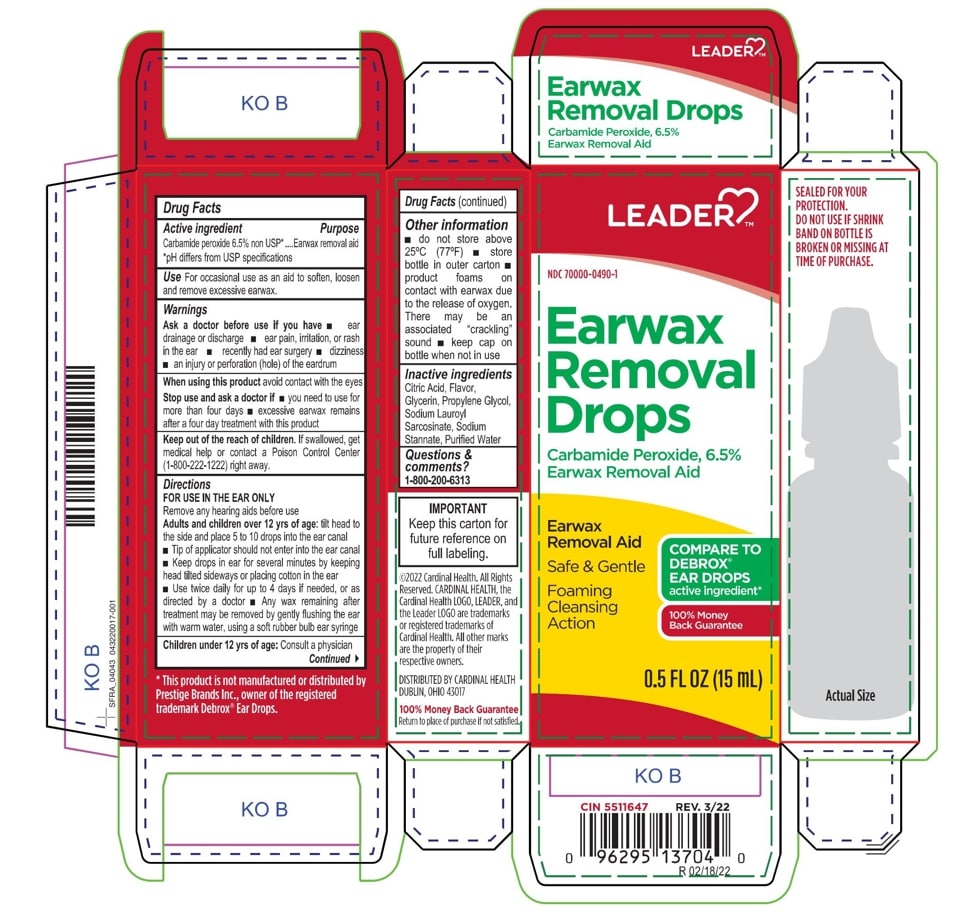
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- INACTIVE INGREDIENT
-
DOSAGE & ADMINISTRATION
- tilt head sideways
- palce 5 to 10 drops into ear
- tip of applicator should not enter ear canal
- keep drops in ear for several minutes by keeping head tilted or placing cotton in the ear
- use twice daily for up to four days if needed, or as directed by a doctor
- any wax remaining after treatment may be removed by gently flushing ear with warm water, using a soft rubber bulb ear syringe
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LEADER EARWAX REMOVAL KIT
carbamide peroxide 6.50% kit liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0490 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBAMIDE PEROXIDE (UNII: 31PZ2VAU81) (HYDROGEN PEROXIDE - UNII:BBX060AN9V) CARBAMIDE PEROXIDE 6.5 g in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM LAUROYL SARCOSINATE (UNII: 632GS99618) WATER (UNII: 059QF0KO0R) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) SODIUM STANNATE (UNII: NJ7C1V83KG) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0490-2 15 mL in 1 CONTAINER; Type 0: Not a Combination Product 05/06/2019 2 NDC:70000-0490-1 1 in 1 CARTON 05/06/2019 2 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M014 05/06/2019 Labeler - Cardinal Health, 110 dba Leader (063997360) Registrant - Pharma Nobis, LLC (118564114) Establishment Name Address ID/FEI Business Operations Pharma Nobis, LLC 118564114 pack(70000-0490) , label(70000-0490) , manufacture(70000-0490) , analysis(70000-0490)