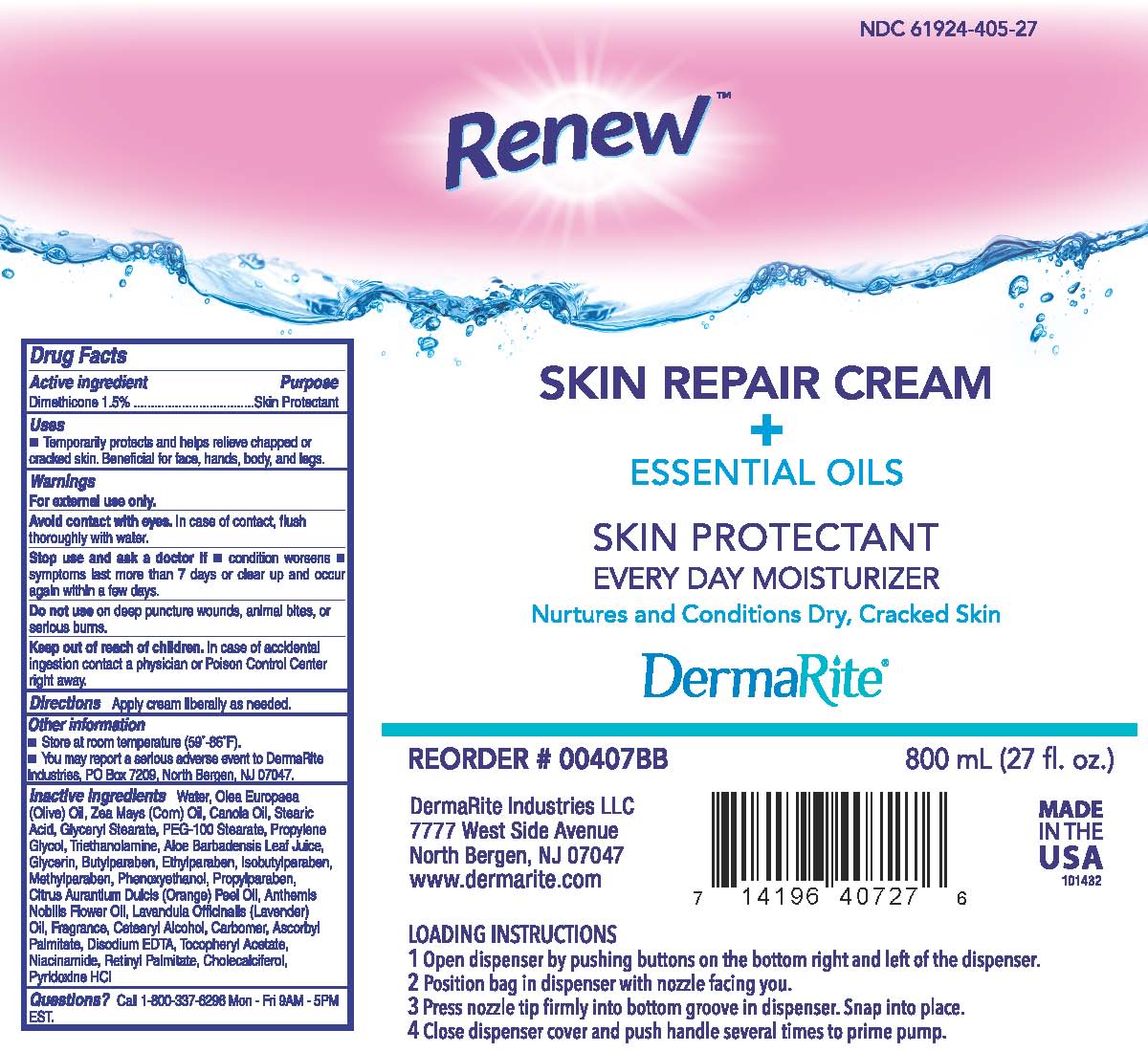
Label: RENEW SKIN REPAIR- otc skin protectant drug products cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 61924-405-04, 61924-405-05, 61924-405-27, 61924-405-32 - Packager: Dermarite Industries LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 17, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient:
- Purpose:
- Uses:
-
Warnings:
- For external use only.
- Avoid contact with eyes. In case of contact, flush thoroughly with water.
- Stop use and ask a doctor if condition worsens or does not improve within 7 days and if symptoms clear up and occur again within a few days.
- Do not use on deep or punctured wounds, animal bites, or serius burns.
- Warnings:
- Directions:
- Other Information:
-
Inactive Infredients:
Water, Olea Europaea(Olive) Oil, Zea Mays (Corn) Oil, Canola Oil, Stearic Acid, glyceryl Stearate, PEG-100 Stearate, Propylene Glycol, Triethanolamine, Aloe Barbadensis Leaf Juice, Glycerin, Butylparaben, Ethylparaben, Isobutylparaben, methylparaben, Phenoxyethanol, propylparaben, Citrus Aurantium Dulcis (orange) Peel Oil, Anthemis Nobilis Flower Oil, Lavandula Officinalis (lavender) Oil, Fragrance, Cetearyl Alcohol, Carbomer, Ascorbyl Palmitate, Disodium EDTA, Tocopheryl Acetate, Niacinamide, Retinyl Palmitate, Cholecalciferol, Pyridoxine HCL
- Questions?
- Package Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
RENEW SKIN REPAIR
otc skin protectant drug products creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61924-405 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 0.015 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ASCORBYL PALMITATE (UNII: QN83US2B0N) BUTYLPARABEN (UNII: 3QPI1U3FV8) CANOLA OIL (UNII: 331KBJ17RK) CARBOMER 940 (UNII: 4Q93RCW27E) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CHOLECALCIFEROL (UNII: 1C6V77QF41) ETHYLPARABEN (UNII: 14255EXE39) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOBUTYLPARABEN (UNII: 0QQJ25X58G) METHYLPARABEN (UNII: A2I8C7HI9T) NIACINAMIDE (UNII: 25X51I8RD4) OLIVE OIL (UNII: 6UYK2W1W1E) PEG-100 STEARATE (UNII: YD01N1999R) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) STEARIC ACID (UNII: 4ELV7Z65AP) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TROLAMINE (UNII: 9O3K93S3TK) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) CORN OIL (UNII: 8470G57WFM) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61924-405-27 793 g in 1 BAG; Type 0: Not a Combination Product 03/03/2011 2 NDC:61924-405-04 113 g in 1 TUBE; Type 0: Not a Combination Product 03/03/2011 3 NDC:61924-405-32 907 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 03/03/2011 4 NDC:61924-405-05 5 g in 1 PACKET; Type 0: Not a Combination Product 03/03/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 03/03/2011 Labeler - Dermarite Industries LLC (883925562) Registrant - Dermarite Industries LLC (883925562) Establishment Name Address ID/FEI Business Operations Dermarite Industries LLC 883925562 manufacture(61924-405)