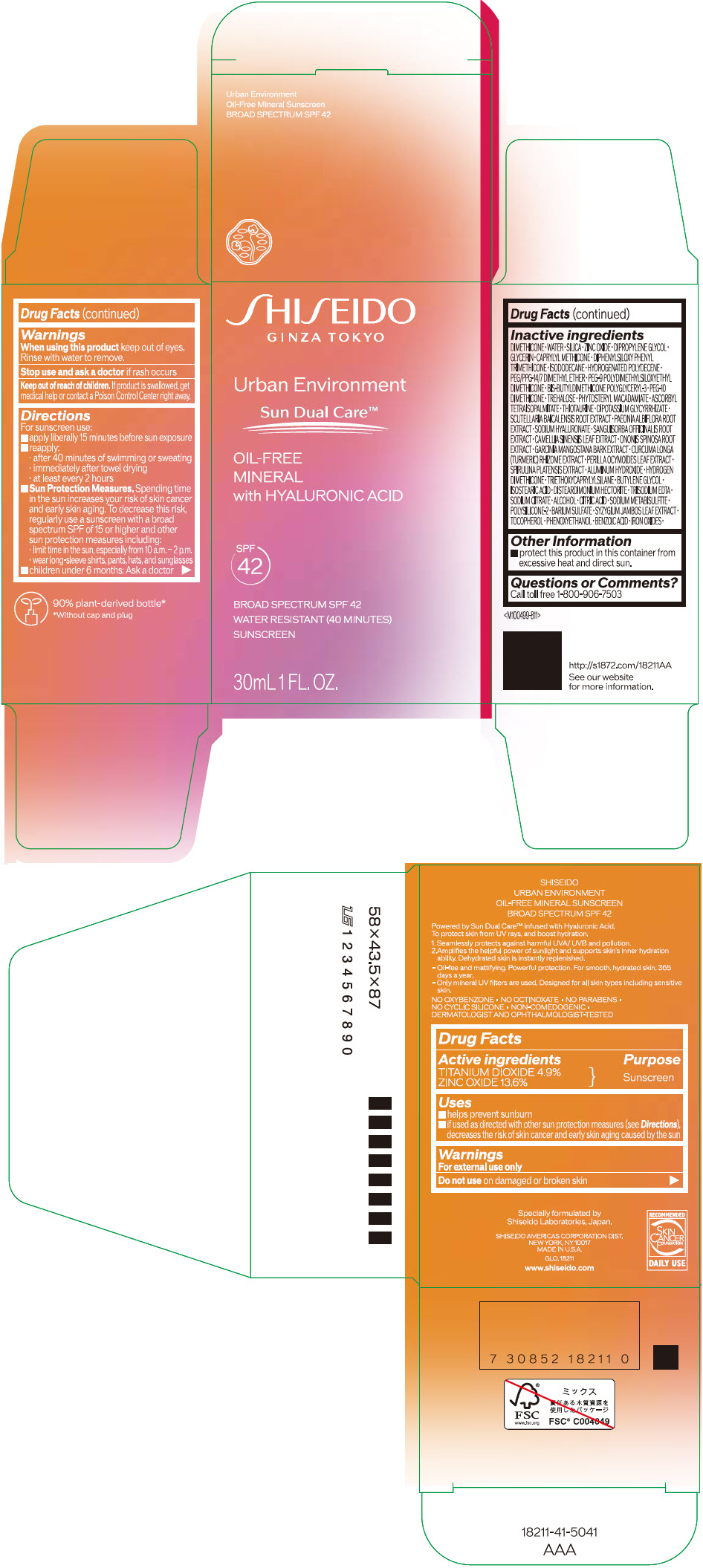
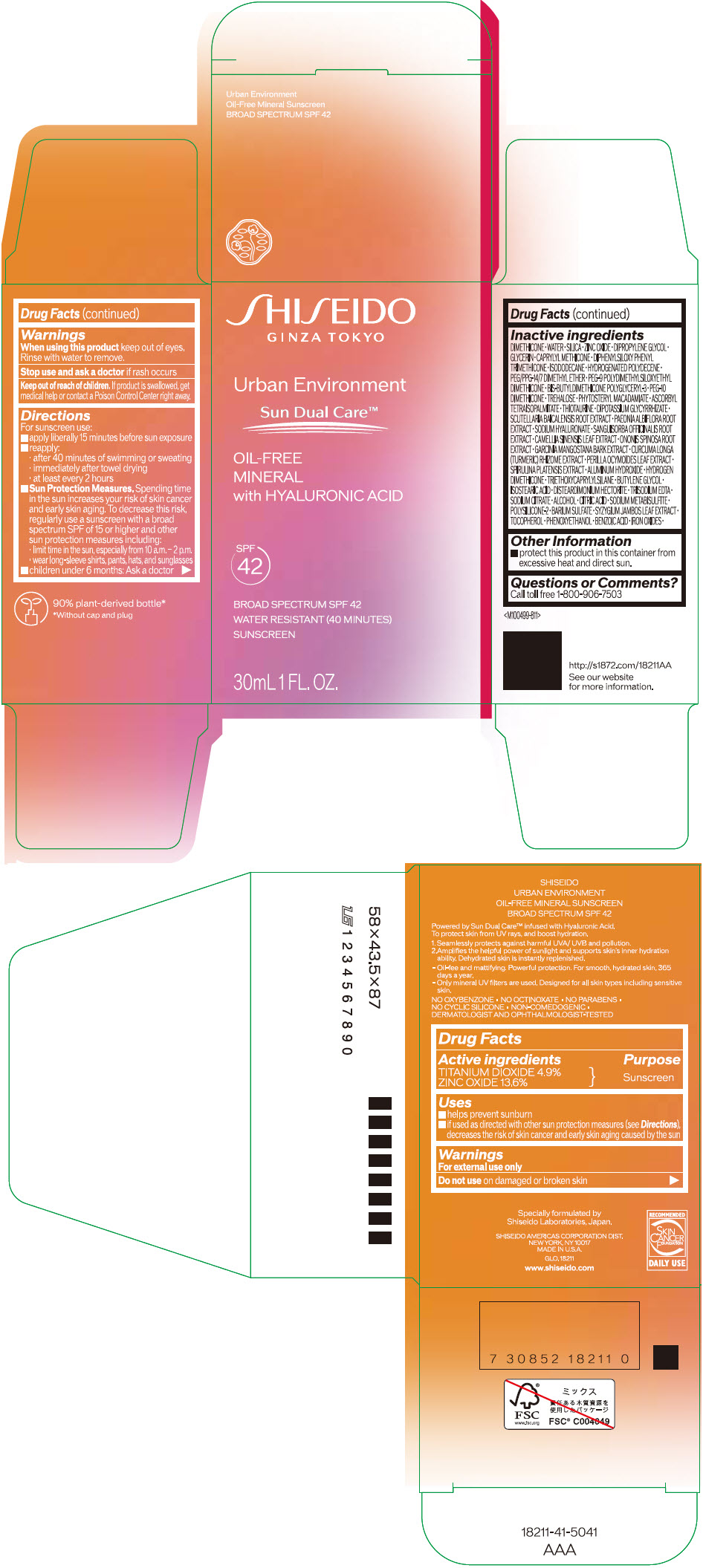
Label: SHISEIDO URBAN ENVIRONMENT OIL-FREE MINERAL SUNSCREEN SPF 42- titanium dioxide and zinc oxide emulsion
- NDC Code(s): 58411-672-10
- Packager: SHISEIDO AMERICAS CORPORATION
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 15, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply:
- after 40 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
-
Inactive Ingredients
DIMETHICONE • WATER • SILICA • ZINC OXIDE • DIPROPYLENE GLYCOL • GLYCERIN • CAPRYLYL METHICONE • DIPHENYLSILOXY PHENYL TRIMETHICONE • ISODODECANE • HYDROGENATED POLYDECENE • PEG/PPG-14/7 DIMETHYL ETHER • PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE • BIS-BUTYLDIMETHICONE POLYGLYCERYL-3 • PEG-10 DIMETHICONE • TREHALOSE • PHYTOSTERYL MACADAMIATE • ASCORBYL TETRAISOPALMITATE • THIOTAURINE • DIPOTASSIUM GLYCYRRHIZATE • SCUTELLARIA BAICALENSIS ROOT EXTRACT • PAEONIA ALBIFLORA ROOT EXTRACT • SODIUM HYALURONATE • SANGUISORBA OFFICINALIS ROOT EXTRACT • CAMELLIA SINENSIS LEAF EXTRACT • ONONIS SPINOSA ROOT EXTRACT • GARCINIA MANGOSTANA BARK EXTRACT • CURCUMA LONGA (TURMERIC) RHIZOME EXTRACT • PERILLA OCYMOIDES LEAF EXTRACT • SPIRULINA PLATENSIS EXTRACT • ALUMINUM HYDROXIDE • HYDROGEN DIMETHICONE • TRIETHOXYCAPRYLYLSILANE • BUTYLENE GLYCOL • ISOSTEARIC ACID • DISTEARDIMONIUM HECTORITE • TRISODIUM EDTA • SODIUM CITRATE • ALCOHOL • CITRIC ACID • SODIUM METABISULFITE • POLYSILICONE-2 • BARIUM SULFATE • SYZYGIUM JAMBOS LEAF EXTRACT • TOCOPHEROL • PHENOXYETHANOL • BENZOIC ACID • IRON OXIDES •
- Other information
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 30 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
SHISEIDO URBAN ENVIRONMENT OIL-FREE MINERAL SUNSCREEN SPF 42
titanium dioxide and zinc oxide emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-672 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 1.8 g in 30 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 30 mL Inactive Ingredients Ingredient Name Strength DIMETHICONE (UNII: 92RU3N3Y1O) WATER (UNII: 059QF0KO0R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DIPROPYLENE GLYCOL (UNII: E107L85C40) GLYCERIN (UNII: PDC6A3C0OX) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) DIPHENYLSILOXY PHENYL TRIMETHICONE (UNII: I445L28B12) ISODODECANE (UNII: A8289P68Y2) HYDROGENATED POLYDECENE TYPE I (UNII: U333RI6EB7) PEG/PPG-14/7 DIMETHYL ETHER (UNII: 6DNW9T7YT2) PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: TYP81E471F) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) TREHALOSE (UNII: B8WCK70T7I) PHYTOSTERYL MACADAMIATE (UNII: 233VSF903M) ASCORBYL TETRAISOPALMITATE (UNII: 47143LT58A) THIOTAURINE (UNII: NQZ2D7AO62) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) SCUTELLARIA BAICALENSIS ROOT (UNII: 7J95K7ID2S) PAEONIA LACTIFLORA ROOT (UNII: 3Z3866YW6P) HYALURONATE SODIUM (UNII: YSE9PPT4TH) SANGUISORBA OFFICINALIS ROOT (UNII: 4NYV2HT01X) GREEN TEA LEAF (UNII: W2ZU1RY8B0) ONONIS SPINOSA ROOT (UNII: FD2FMC53M1) GARCINIA MANGOSTANA BARK (UNII: N6A4WY566I) TURMERIC (UNII: 856YO1Z64F) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) HYDROGEN DIMETHICONE (13 CST) (UNII: 4QGR4P2YOI) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ISOSTEARIC ACID (UNII: X33R8U0062) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) EDETATE TRISODIUM (UNII: 420IP921MB) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) ALCOHOL (UNII: 3K9958V90M) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM METABISULFITE (UNII: 4VON5FNS3C) BARIUM SULFATE (UNII: 25BB7EKE2E) SYZYGIUM JAMBOS LEAF (UNII: 407Z4W5LFF) TOCOPHEROL (UNII: R0ZB2556P8) PHENOXYETHANOL (UNII: HIE492ZZ3T) BENZOIC ACID (UNII: 8SKN0B0MIM) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-672-10 1 in 1 CARTON 01/01/2022 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH DRUG M020 01/01/2022 Labeler - SHISEIDO AMERICAS CORPORATION (193691821) Establishment Name Address ID/FEI Business Operations Shiseido America Inc. 782677132 ANALYSIS(58411-672) , MANUFACTURE(58411-672)