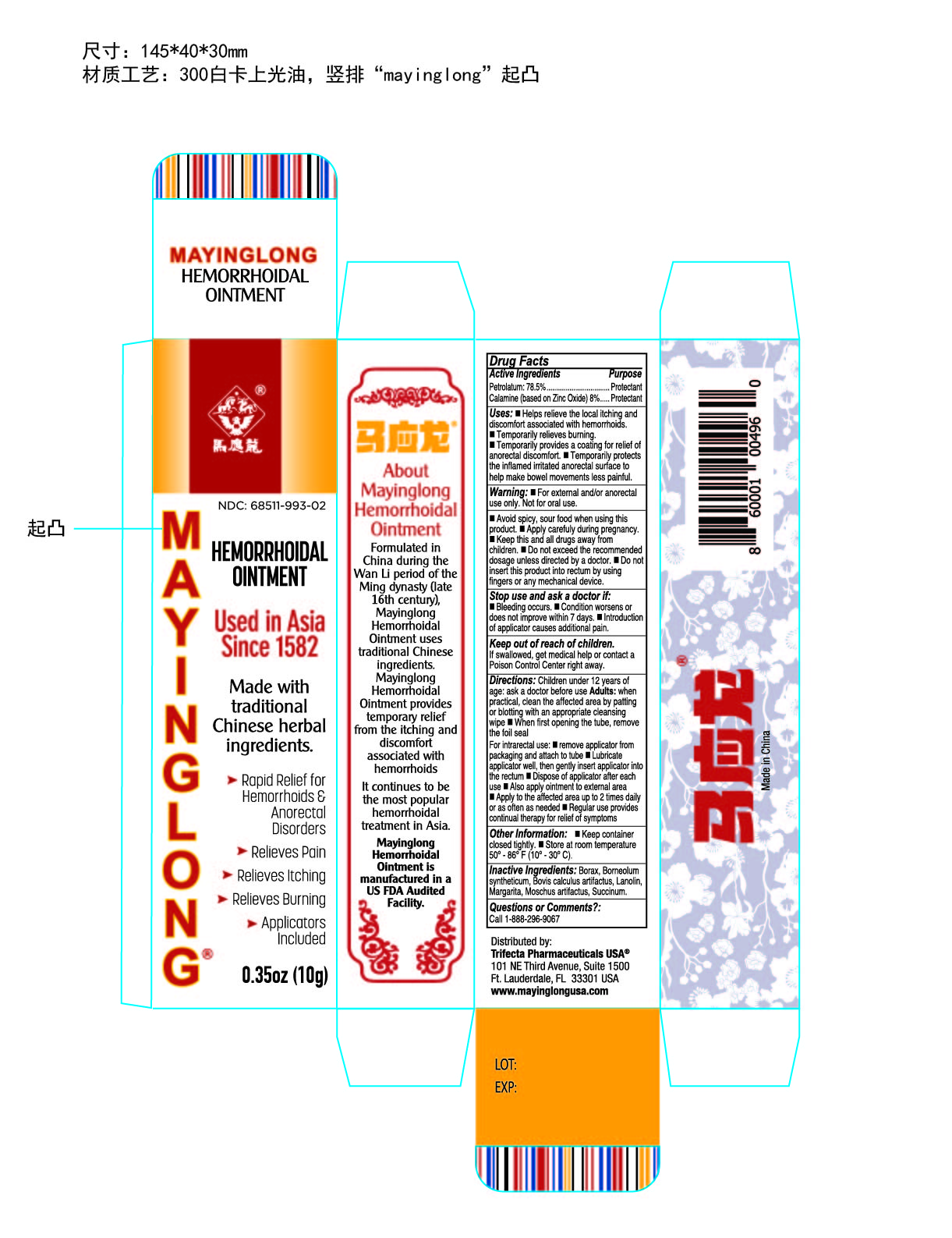
Label: MAYINGLONG HEMORRHOIDS- zinc oxide, petrolatum, lanolin ointment
-
Contains inactivated NDC Code(s)
NDC Code(s): 68511-993-02, 68511-993-03, 68511-993-04 - Packager: Mayinglong Pharmaceutical Group Co Ltd
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 19, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
-
Uses
helps relieve the local itching and discomfort associated with hemorrhoids
temporarily shrinks hemorrhoidal tissue and relieves burning
temporarily provides a coating for relief of anorectal discomforts
temporarily protects the inflamed, irritated anorectal surface to help make bowel movements less painful
- Warning
- Stop use
- Warning
- Warning
- Direction
- Other information
- Inactive ingredients
- Drug Facts
-
INGREDIENTS AND APPEARANCE
MAYINGLONG HEMORRHOIDS
zinc oxide, petrolatum, lanolin ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68511-993 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 800 mg in 10000 mg PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 7850 mg in 10000 mg Inactive Ingredients Ingredient Name Strength MUSK KETONE (UNII: 483V3E1L6J) PEARL (HYRIOPSIS CUMINGII) (UNII: A75L5FZ40U) BORNEOL (UNII: M89NIB437X) SODIUM BORATE (UNII: 91MBZ8H3QO) LANOLIN (UNII: 7EV65EAW6H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68511-993-02 10000 mg in 1 CARTON; Type 0: Not a Combination Product 10/25/2017 2 NDC:68511-993-03 4000 mg in 1 CARTON; Type 0: Not a Combination Product 08/19/2019 3 NDC:68511-993-04 20000 mg in 1 CARTON; Type 0: Not a Combination Product 08/19/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part346 12/15/2003 Labeler - Mayinglong Pharmaceutical Group Co Ltd (526823828) Registrant - Mayinglong Pharmaceutical Group Co Ltd (526823828) Establishment Name Address ID/FEI Business Operations Mayinglong Pharmaceutical Group Co Ltd 526823828 manufacture(68511-993)