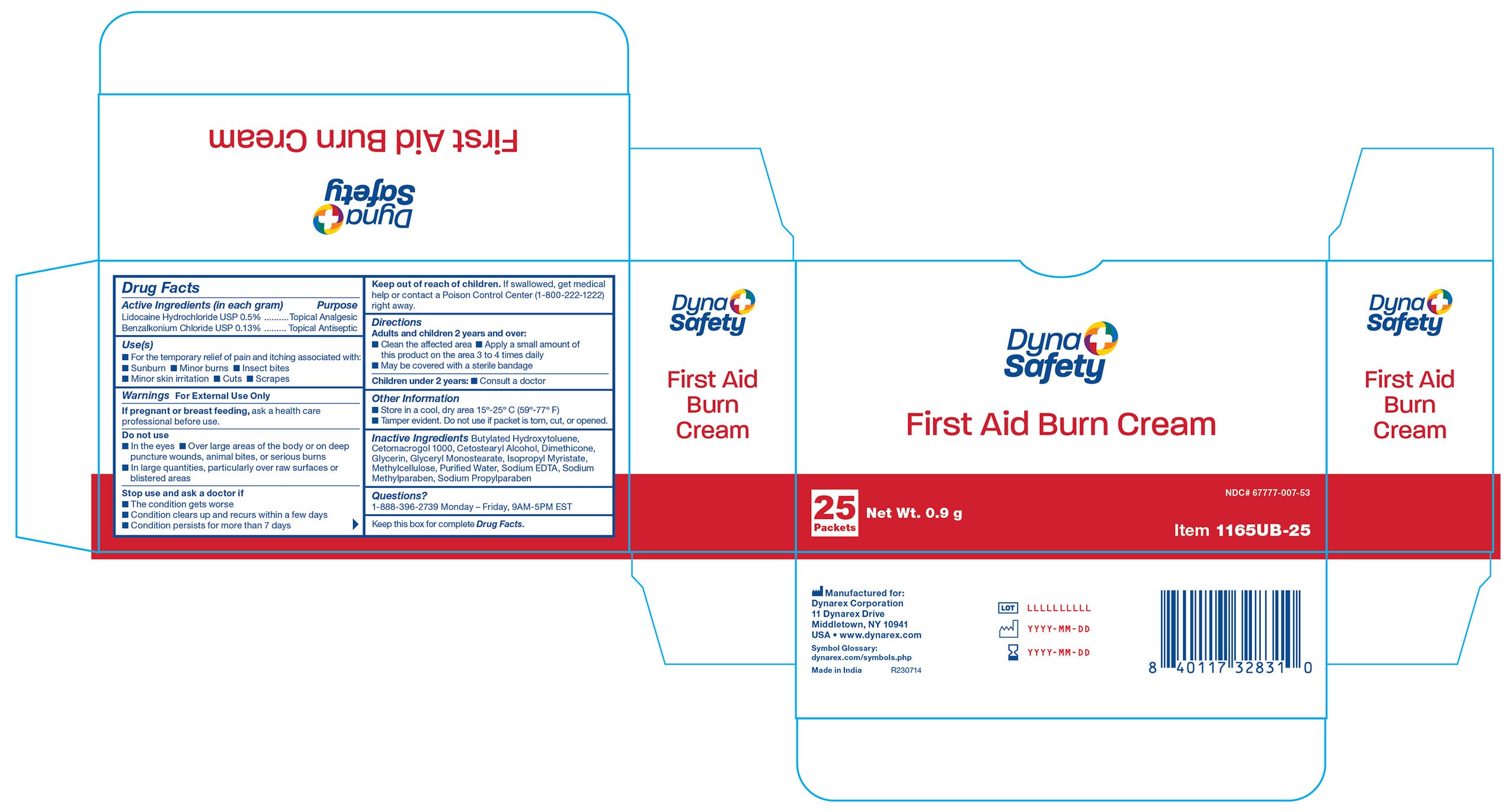
Label: BURN FIRST AID cream
-
NDC Code(s):
67777-007-49,
67777-007-50,
67777-007-51,
67777-007-52, view more67777-007-53, 67777-007-54
- Packager: Dynarex Corporation
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 22, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- in the eyes
- over large areas of the body or on deep puncture wounds, animal bites, or serious burns
- in large quantities, particularly over raw surfaces or blistered areas
- Directions
- Other Information
- Inactive Ingredients
- 1165 Label
- 1165UB-10 Label
- 1165UB-25 Label
-
INGREDIENTS AND APPEARANCE
BURN FIRST AID
burn first aid creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67777-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 50 mg in 10000 mg BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 13 mg in 10000 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) EDETATE SODIUM (UNII: MP1J8420LU) METHYLPARABEN SODIUM (UNII: CR6K9C2NHK) PROPYLPARABEN SODIUM (UNII: 625NNB0G9N) CETETH-20 (UNII: I835H2IHHX) METHYLCELLULOSE (4000 CPS) (UNII: MRJ667KA5E) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) DIMETHICONE 350 (UNII: 2Y53S6ATLU) Product Characteristics Color Score Shape FREEFORM Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67777-007-50 1728 in 1 CASE 09/17/2018 1 NDC:67777-007-49 144 in 1 BOX 1 900 mg in 1 PACKET; Type 0: Not a Combination Product 2 NDC:67777-007-52 1000 in 1 CASE 09/17/2018 2 NDC:67777-007-51 10 in 1 BOX 2 900 mg in 1 PACKET; Type 0: Not a Combination Product 3 NDC:67777-007-54 1800 in 1 CASE 09/17/2018 3 NDC:67777-007-53 25 in 1 BOX 3 900 mg in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 09/17/2018 Labeler - Dynarex Corporation (008124539)



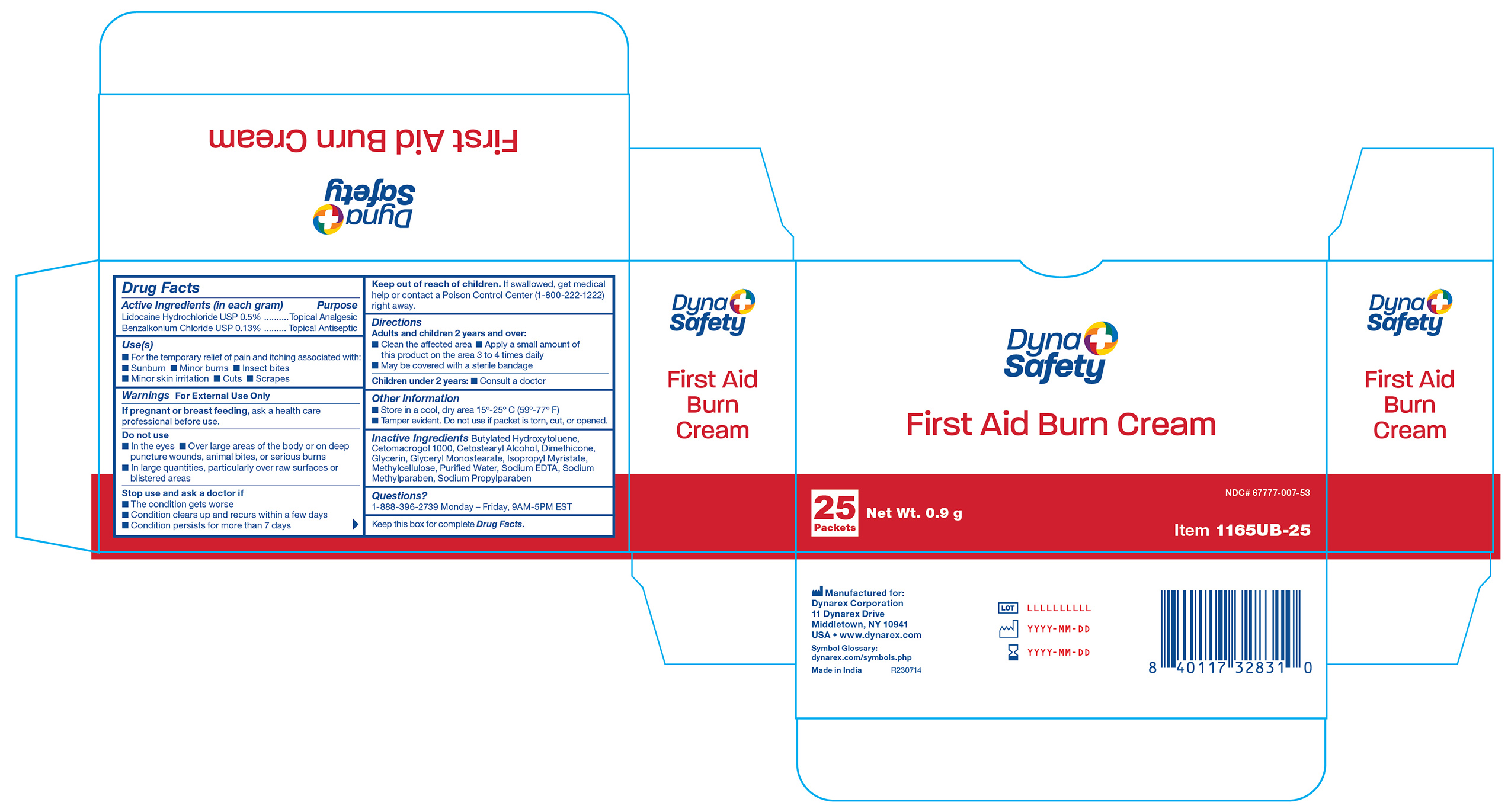

 1165UB-10
1165UB-10