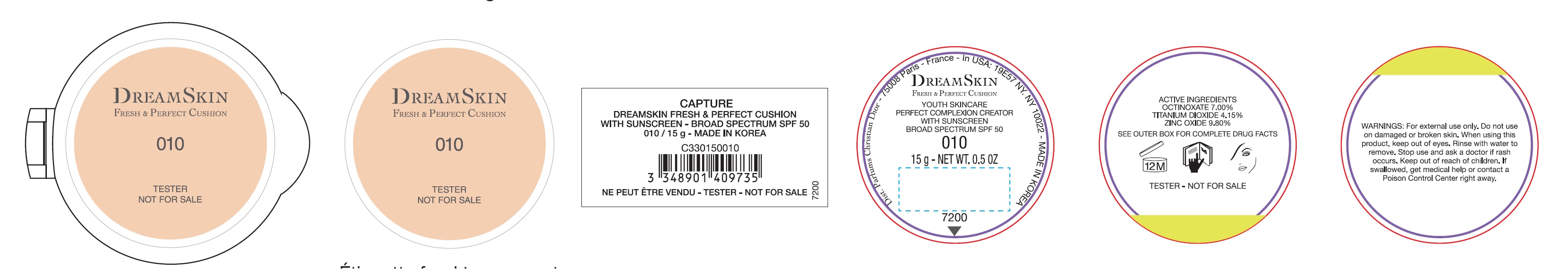
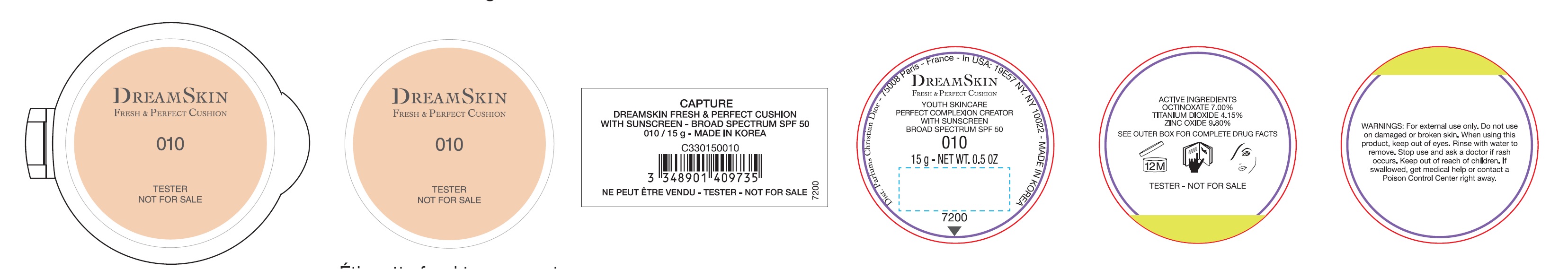
Label: CAPTURE DREAMSKIN FRESH AND PERFECT CUSHION WITH SUNSCREEN BROAD SPECTRUM SPF 50 010- octinoxate,titanium dioxide,zinc oxide emulsion
- NDC Code(s): 61957-2300-0, 61957-2300-1, 61957-2300-2
- Packager: Parfums Christian Dior
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 3, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure.
- Reapply at least every 2 hours
- Use a water resistant sunscreen if swimming or sweating
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses.
- Children under 6 months of age: ask a docto.
. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures Sun Protection Measures
including:
- Other information
-
Inactive ingredients:
AQUA (WATER)• CYCLOPENTASILOXANE• CYCLOHEXASILOXANE• PEG-10 DIMETHICONE•DIMETHICONE •BUTYLENE GLYCOL DICAPRYLATE/DICAPRATE• PROPANEDIOL •LAURYLPEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE• HDVTRIMETHYLOL HEXYLLACTONE CROSSPOLYMER TRISILOXANE •PHENYL TRIMETHICONE• POLYMETHYL METHACRYLATE•GLYCERIN •SODIUM CHLORIDE• DIMETHICONE/PEG-10/15 CROSSPOLYMER •ALUMINUM HYDROXDE• DISTEARDIMONIUM HECTORITE •PHENOXYETHANOL •STEARIC ACID• XYLITOL •TRIETHOXYCAPRYLYLSILANE• PARFUM (FRAGRANCE) •CAPRYLYL GLYCOL•ETHYLHEXYLGLYCERIN •DISODIUM EDTA •SILICA •ZINC GLUCONATE• ADENOSINE•SYNTHETNC FLUORPHLOGOPITE •PALMARIA PALMAIA EXTRACT•LIMONENE•SODIUMBENZOATE•GERANIOL •ALPHA-ISOMETHYL IONONE• BUTYLENE GLYCOL• JASMINUM OFFICINALE (JASMINE) FLOWER EXTRACT• CITRONELLOL• ALCOHOL• TOCOPHEROL• HYDROGEN DIMETHICONE•SAPONINS• AFRAMOMUM ANGUSTIFOLIUM SEED EXTRACT.[MAY CONTAIN: CI 77491, CI77492, CI 77499 (IRON OXIDES) •CI 77891 (TITANIUM DIOXIDE)].
- Package Labeling(2*15g)
- Package Labeling(TESTER 15g)
-
INGREDIENTS AND APPEARANCE
CAPTURE DREAMSKIN FRESH AND PERFECT CUSHION WITH SUNSCREEN BROAD SPECTRUM SPF 50 010
octinoxate,titanium dioxide,zinc oxide emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61957-2300 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.07 mg in 1 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 0.0415 mg in 1 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 0.098 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) CYCLOMETHICONE 6 (UNII: XHK3U310BA) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) DIMETHICONE (UNII: 92RU3N3Y1O) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PROPANEDIOL (UNII: 5965N8W85T) LAURYL PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: 25G622K2RA) HEXAMETHYLENE DIISOCYANATE/TRIMETHYLOL HEXYLLACTONE CROSSPOLYMER (UNII: WB5K9Y35Y9) TRISILOXANE (UNII: 9G1ZW13R0G) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) GLYCERIN (UNII: PDC6A3C0OX) SODIUM CHLORIDE (UNII: 451W47IQ8X) DIMETHICONE/PEG-10/15 CROSSPOLYMER (UNII: 21AS8B1BSS) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) PHENOXYETHANOL (UNII: HIE492ZZ3T) STEARIC ACID (UNII: 4ELV7Z65AP) XYLITOL (UNII: VCQ006KQ1E) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) CAPRYLYL GLYCOL (UNII: 00YIU5438U) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ADENOSINE (UNII: K72T3FS567) PALMARIA PALMATA (UNII: 7832HOY4ZQ) LIMONENE, (+)- (UNII: GFD7C86Q1W) SODIUM BENZOATE (UNII: OJ245FE5EU) GERANIOL (UNII: L837108USY) ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) JASMINUM OFFICINALE FLOWER (UNII: 0Q8K841432) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) ALCOHOL (UNII: 3K9958V90M) TOCOPHEROL (UNII: R0ZB2556P8) AFRAMOMUM ANGUSTIFOLIUM SEED (UNII: OSF83Q896J) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61957-2300-0 2 in 1 PACKAGE 11/26/2020 1 15 g in 1 JAR; Type 0: Not a Combination Product 2 NDC:61957-2300-1 15 g in 1 JAR; Type 0: Not a Combination Product 11/26/2020 3 NDC:61957-2300-2 15 g in 1 JAR; Type 0: Not a Combination Product 11/26/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/26/2020 Labeler - Parfums Christian Dior (275252245)