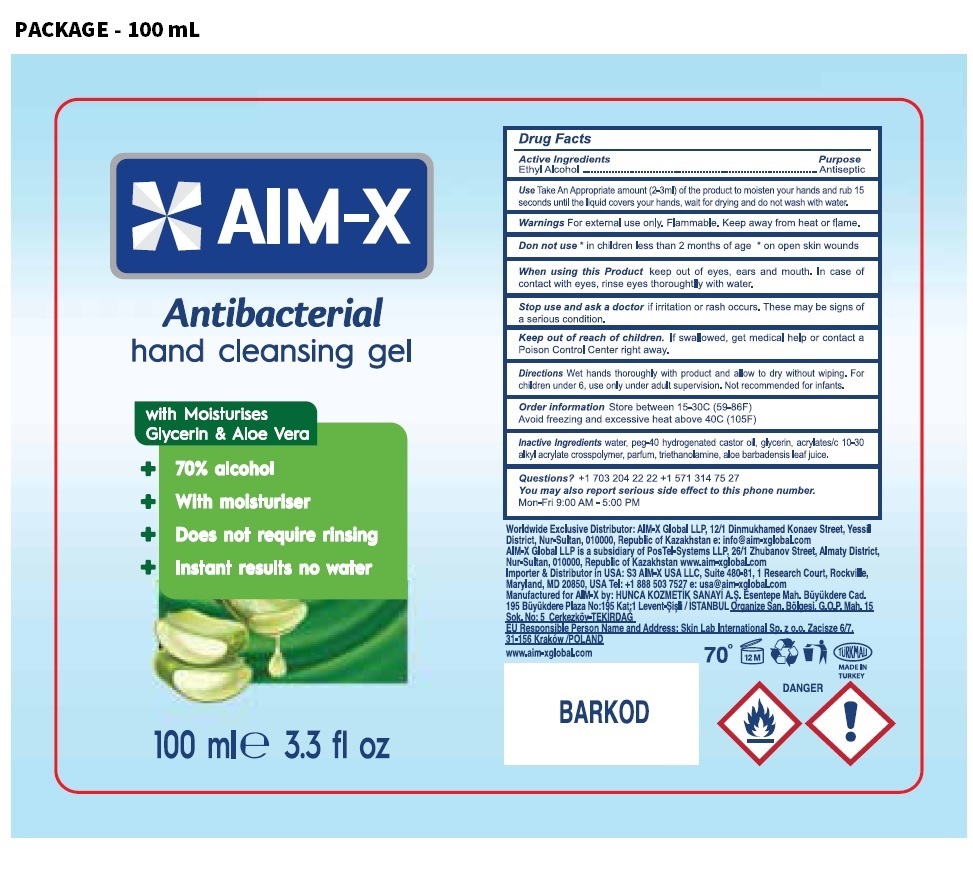
Label: AIM-X ANTIBACTERIAL HAND CLEANSING GEL- ethyl alcohol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 81410-101-01, 81410-101-02, 81410-101-03, 81410-101-04, view more81410-101-05, 81410-101-06, 81410-101-07, 81410-101-08, 81410-101-09, 81410-101-10, 81410-101-11, 81410-101-12, 81410-101-13, 81410-101-14, 81410-101-15, 81410-101-16, 81410-101-17, 81410-101-18, 81410-101-19, 81410-101-20, 81410-101-21, 81410-101-22, 81410-101-23, 81410-101-24, 81410-101-25, 81410-101-26, 81410-101-27, 81410-101-28 - Packager: AIM-X GLOBAL, TOO.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 15, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Purpose
- Use
-
Warnings
For external use only. Flammable. Keep away from heat or flame.
Do not use * in children less than 2 months of age * on open skin wounds
When using this Product keep out of eyes, ears and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
- Directions
- Other information
- Inactive Ingredients
- Questions?
-
SPL UNCLASSIFIED SECTION
with Moisturises
Glycerin & Aloe Vera
+ 70% alcohol
+ with moisturiser
+ Does not require rinsing
+ Instant results no water
Worldwide Exclusive Distributor: AIM-X Global LLP, 12/1 Dinmukhamed Konaev Street, Yessil District, Nur-Sultan, 010000, Republic of Kazakhstan
e: info@aim-xglobal.com
AIM-X Global LLP is a subsidiary of PosTel-Systems LLP, 26/1 Zhubanov Street, Almaty District, Nur-Sultan, 010000, Republic of Kazakhstan www.aim-xglobal.com
Importer & Distributor in USA: S3 AIM-X USA LLC, Suite 480-81, 1 Research Court, Rockville, Maryland, MD 20850,
USA Tel: +1 888 503 7527 e: usa@aim-xglobal.com
Manufactured for AIM-X by: HUNCA KOZMETIK SANAYI A.S. Esentepe Mah. Büyükdere Cad. 195 Büyükdere Plaza No: 195 Kat:1
Levent-Sisli/ ISTANBUL Organize San. Bölgesi, G.O.P. Mah. 15 Sok. No: 5 Cerkezköy-TEKIRDAG
EU Responsible Person Name and Address: Skin Lab International Sp.z o.o. Zacisze 6/7, 31-156 Kraków /POLAND
www.aim-xglobal.com
TÜRKMALI
MADE IN TURKEY
- Packaging
-
INGREDIENTS AND APPEARANCE
AIM-X ANTIBACTERIAL HAND CLEANSING GEL
ethyl alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81410-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) GLYCERIN (UNII: PDC6A3C0OX) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) TROLAMINE (UNII: 9O3K93S3TK) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81410-101-01 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 2 NDC:81410-101-02 7 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 3 NDC:81410-101-03 10 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 4 NDC:81410-101-04 20 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 5 NDC:81410-101-05 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 6 NDC:81410-101-06 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 7 NDC:81410-101-07 75 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 8 NDC:81410-101-08 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 9 NDC:81410-101-09 120 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 10 NDC:81410-101-10 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 11 NDC:81410-101-11 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 12 NDC:81410-101-12 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 13 NDC:81410-101-13 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 14 NDC:81410-101-14 400 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 15 NDC:81410-101-15 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 16 NDC:81410-101-16 650 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 17 NDC:81410-101-17 750 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 18 NDC:81410-101-18 900 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 19 NDC:81410-101-19 1000 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 20 NDC:81410-101-20 1250 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 21 NDC:81410-101-21 1500 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 22 NDC:81410-101-22 2000 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 23 NDC:81410-101-23 2500 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/25/2020 24 NDC:81410-101-24 5000 mL in 1 CAN; Type 0: Not a Combination Product 11/25/2020 25 NDC:81410-101-25 10000 mL in 1 CAN; Type 0: Not a Combination Product 11/25/2020 26 NDC:81410-101-26 15000 mL in 1 CAN; Type 0: Not a Combination Product 11/25/2020 27 NDC:81410-101-27 20000 mL in 1 CAN; Type 0: Not a Combination Product 11/25/2020 28 NDC:81410-101-28 30000 mL in 1 CAN; Type 0: Not a Combination Product 11/25/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 11/25/2020 Labeler - AIM-X GLOBAL, TOO. (521191626)