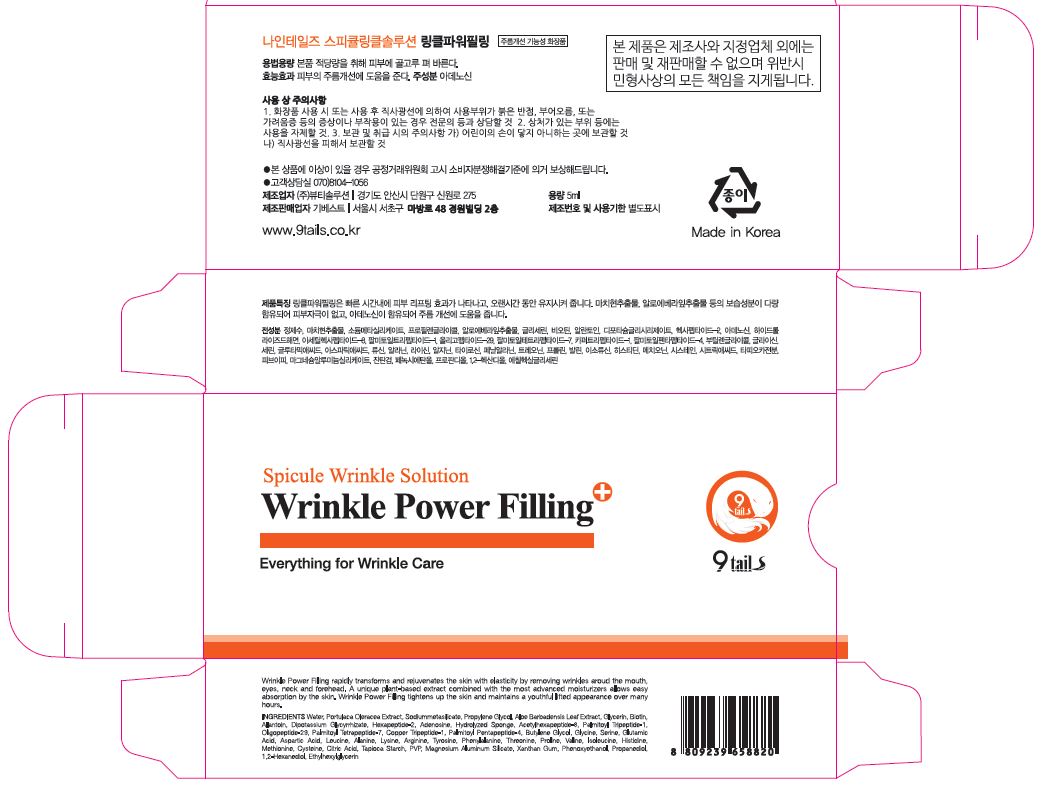
Label: WRINKLE POWER FILLING- glycerin gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 72176-0200-1 - Packager: GIBEST
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 13, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
[How to Use]
- Towel dry after face washing.
- Apply thin layer to the direction of wrinkle. Keep patting gently for about 15 seconds.
- Try not to talk or move for at least 2 minutes until dry.
- Strain feeling of your skin will disappear naturally.
- Shake well before you use.
- The nature of Hydrolyzed Sponge, it may be pricking little bit depends on individual's skin condition. However, it is quite natural and proving for working to your skin.
- WARNINGS
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
WRINKLE POWER FILLING
glycerin gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72176-0200 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 3.55 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ADENOSINE (UNII: K72T3FS567) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72176-0200-1 5 mL in 1 TUBE; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part347 03/01/2018 Labeler - GIBEST (689837540) Registrant - GIBEST (689837540) Establishment Name Address ID/FEI Business Operations GIBEST 689837540 manufacture(72176-0200)