Label: TOPCARE COUGH DM- dextromethorphan polistirex suspension
- NDC Code(s): 36800-899-21
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 8, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
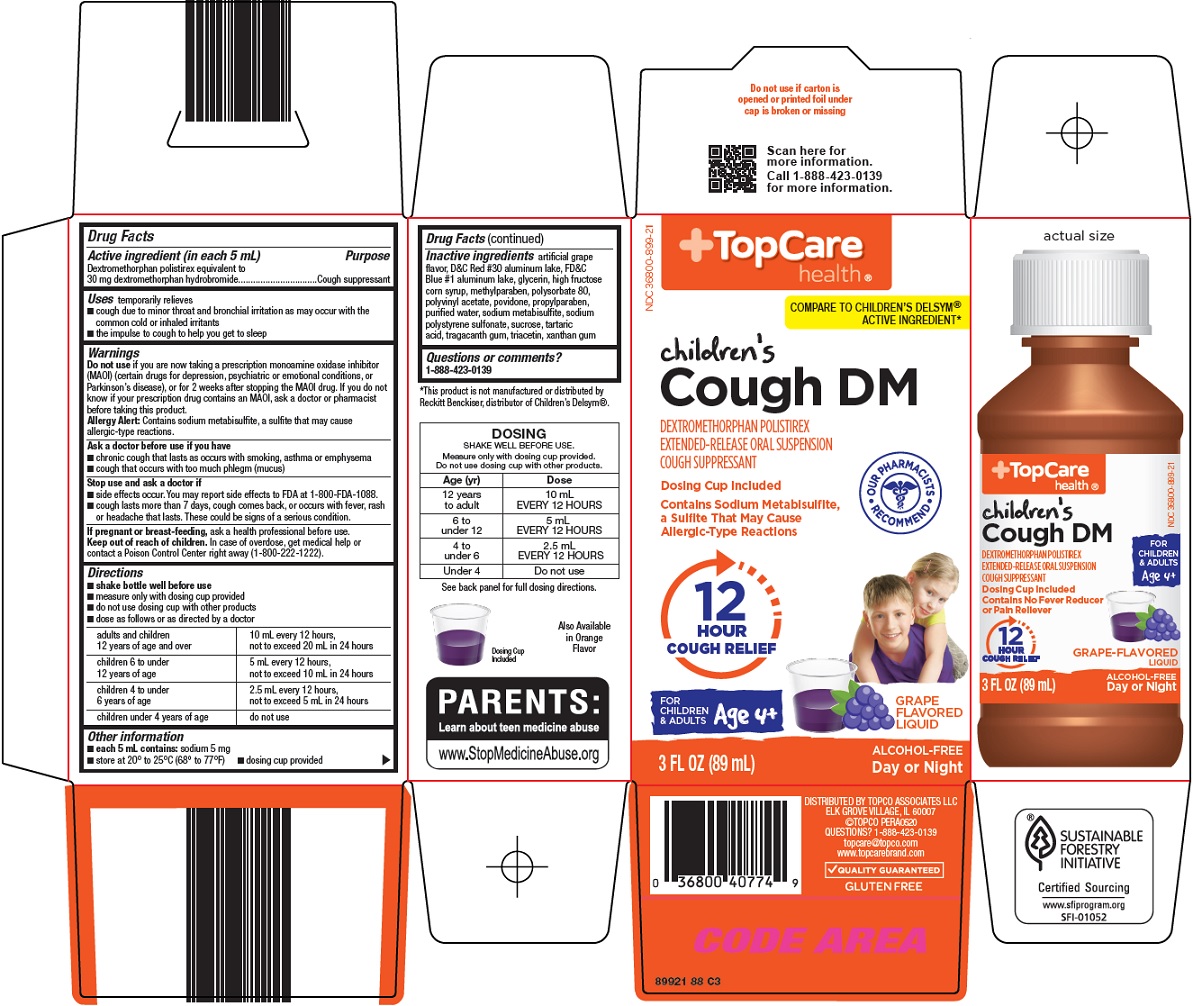
- Active ingredient (in each 5 mL)
- Purpose
- Uses
- Warnings
-
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Allergy Alert: Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions.
- Ask a doctor before use if you have
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- •
- shake bottle well before use
- •
- measure only with dosing cup provided
- •
- do not use dosing cup with other products
- •
- dose as follows or as directed by a doctor
adults and children 12 years of age and over
10 mL every 12 hours, not to exceed 20 mL in 24 hours
children 6 to under 12 years of age
5 mL every 12 hours, not to exceed 10 mL in 24 hours
children 4 to under 6 years of age
2.5 mL every 12 hours, not to exceed 5 mL in 24 hours
children under 4 years of age
do not use
- Other information
-
Inactive ingredients
artificial grape flavor, D&C Red #30 aluminum lake, FD&C Blue #1 aluminum lake, glycerin, high fructose corn syrup, methylparaben, polysorbate 80, polyvinyl acetate, povidone, propylparaben, purified water, sodium metabisulfite, sodium polystyrene sulfonate, sucrose, tartaric acid, tragacanth gum, triacetin, xanthan gum
- Questions or comments?
-
Package/Label Principal Display Panel
COMPARE TO CHILDREN’S DELSYM® ACTIVE INGREDIENT
children’s Cough DM
DEXTROMETHORPHAN POLISTIREX EXTENDED-RELEASE ORAL SUSPENSION COUGH SUPPRESSANT
Dosing Cup Included
OUR PHARMACISTS RECOMMEND
Contains Sodium Metabisulfite, a Sulfite That May Cause Allergic-Type Reactions
12 HOUR COUGH RELIEF
FOR CHILDREN & ADULTS Age 4+
GRAPE FLAVORED LIQUID
3 FL OZ (89 mL)
ALCOHOL-FREE
Day or Night
-
INGREDIENTS AND APPEARANCE
TOPCARE COUGH DM
dextromethorphan polistirex suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-899 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg in 5 mL Inactive Ingredients Ingredient Name Strength D&C RED NO. 30 (UNII: 2S42T2808B) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ACETATE (UNII: 32K497ZK2U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM METABISULFITE (UNII: 4VON5FNS3C) SODIUM POLYSTYRENE SULFONATE (UNII: 1699G8679Z) SUCROSE (UNII: C151H8M554) TARTARIC ACID (UNII: W4888I119H) TRAGACANTH (UNII: 2944357O2O) TRIACETIN (UNII: XHX3C3X673) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color PURPLE Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-899-21 1 in 1 CARTON 07/30/2015 1 89 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091135 07/30/2015 Labeler - Topco Associates LLC (006935977)