Label: BETR REMEDIES CHEST CONGESTION RELIEF- guaifenesin tablet, multilayer, extended release
- NDC Code(s): 80267-010-74
- Packager: Live Betr LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated March 20, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
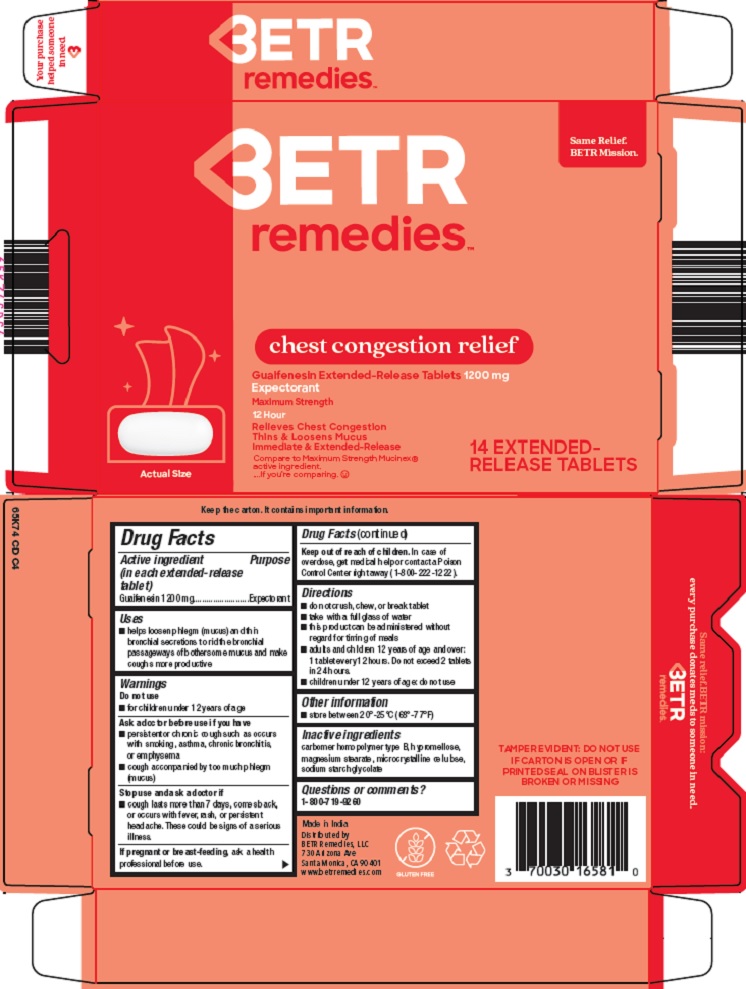
- Active ingredient (in each extended-release tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- •
- cough accompanied by too much phlegm (mucus)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Package/Label Principal Display Panel
BETR remedies™
Same Relief. BETR Mission.
chest congestion relief
Guaifenesin Extended-Release Tablets 1200 mg
Expectorant
Maximum Strength
12 Hour
Relieves Chest Congestion
Thins & Loosens Mucus
Immediate & Extended-Release
Compare to Maximum Strength Mucinex® active ingredient
…. If you ‘re comparing.
14 EXTENDED-RELEASE TABLETS
-
INGREDIENTS AND APPEARANCE
BETR REMEDIES CHEST CONGESTION RELIEF
guaifenesin tablet, multilayer, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80267-010 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL OR ALLYL SUCROSE CROSSLINKED) (UNII: K6MOM3T5YL) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color WHITE Score no score Shape OVAL Size 22mm Flavor Imprint Code G;1200 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80267-010-74 14 in 1 CARTON 03/20/2023 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213420 03/20/2023 Labeler - Live Betr LLC (117423281)