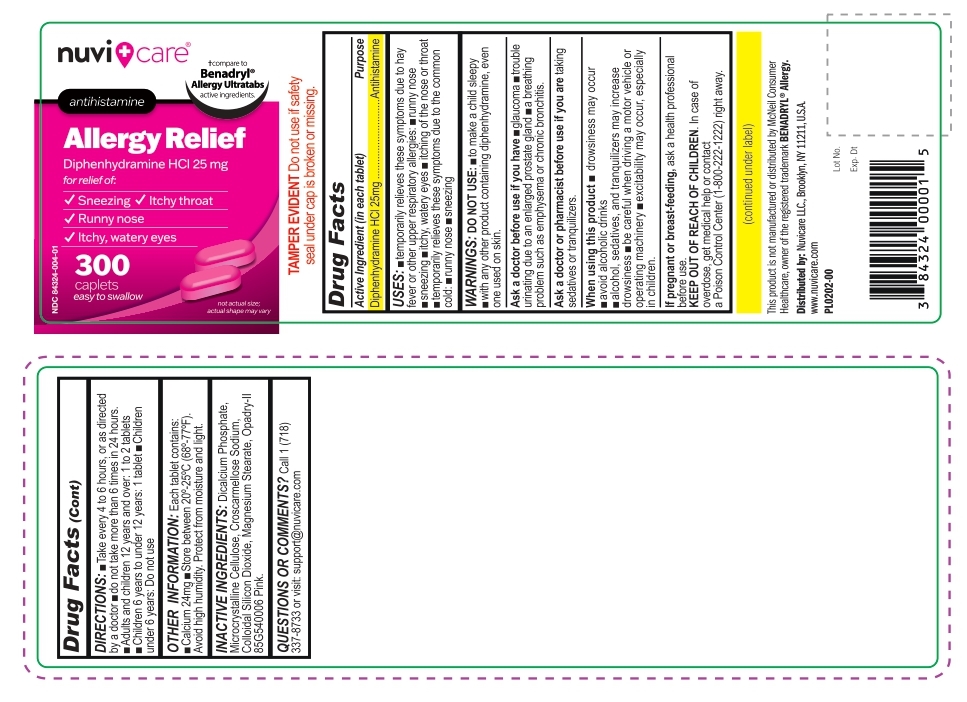
Label: ALLERGY RELIEF- diphenhydramine hcl 25 mg tablet
- NDC Code(s): 84324-004-01
- Packager: NUVICARE LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated September 10, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient (in each tablet)
- Purpose
- Uses:
- Warnings:
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
diphenhydramine hcl 25 mg tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84324-004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color pink Score no score Shape OVAL Size 11mm Flavor Imprint Code S4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84324-004-01 300 in 1 BOTTLE; Type 0: Not a Combination Product 07/17/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 07/17/2024 Labeler - NUVICARE LLC (119257565) Registrant - NUVICARE LLC (119257565)