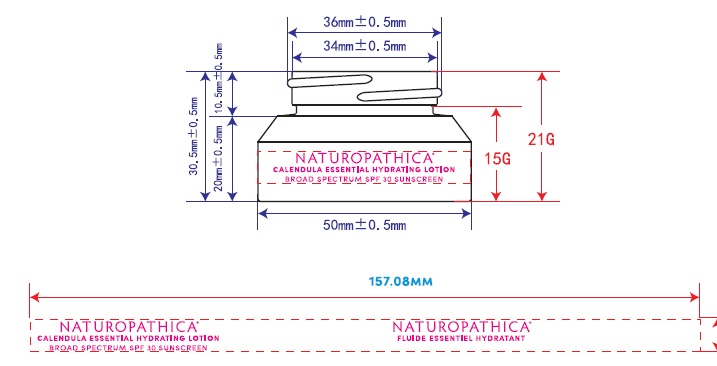
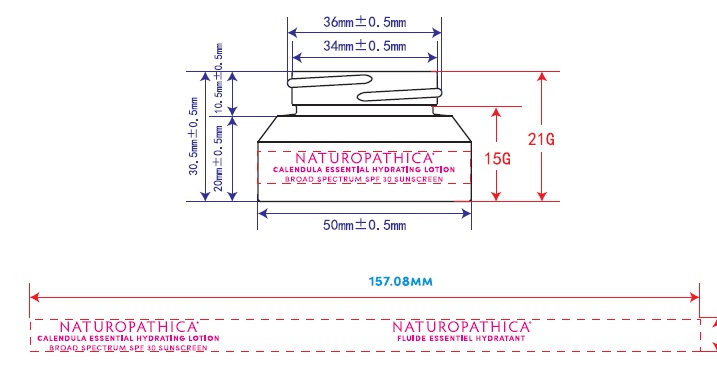
Label: NATUROPATHICA- zinc oxide lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 54111-152-50, 54111-152-51 - Packager: Bentley Laboratories, LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 1, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug FactsActive Ingredient
- Purpose
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
-
Directions
• apply liberally 15 minutes before sun exposure
• reapply at least every two hours
• use a water resistant sunscreen if swimming or sweating
• Sun Protection Measures. Spending time in
the sun increases your risk of skin cancer and
early skin aging. To decrease this risk,
regularly use a sunscreen with a Broad
Spectrum SPF 15 or higher and other
sun protection measures including:
• limit time in the sun, especially from 10a.m. - 2p.m.
• wear long-sleeved shirts, pants, hats and sunglasses
• children under 6 months of age: consult a doctor - Other Information
- QUESTIONS
-
Inactive Ingredients
Aqua/Water/Eau,
Coco-Caprylate/Caprate, C12-15 Alkyl Benzoate,
Galactoarabinan, Glycerin, Triethyl Citrate,
Cetearyl Alcohol, Behenyl Alcohol, Butyloctyl
Salicylate, Propanediol, Cetearyl Glucoside,
Carthamus Tinctorius (Safflower) Oleosomes, Sodium
Stearoyl Glutamate, Lavandula Angustifolia
(Lavender) Oil, Lavandula Angustifolia (Lavender)
Flower/Leaf/Stem Extract, Rosa Damascena Flower
Oil, Tagetes Minuta Flower Oil, Citrus Aurantium Dulcis
(Orange) Flower Oil, Ethyl Ferulate, Undaria
Pinnatifida Extract, Polyglyceryl-3 Polyricinoleate,
Glyceryl Caprylate, Borago Officinalis Seed Oil,
Hydroxyacetophenone, Xylitylglucoside,
Coco-Glucoside, Bisabolol, Xanthan Gum, Isostearic
Acid, Anhydroxylitol, Citric Acid, Xylitol,
Caprylhydroxamic Acid, Polygonum Aviculare Extract,
Potassium Sorbate, Sodium Benzoate, Carthamus
Tinctorius (Safflower) Seed Oil, Calendula Officinalis
Flower Extract, Trehalose, Xanthophylls
- Product Label
-
INGREDIENTS AND APPEARANCE
NATUROPATHICA
zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54111-152 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 15.002 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) COCO-CAPRYLATE/CAPRATE (UNII: 8D9H4QU99H) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) GALACTOARABINAN (UNII: SL4SX1O487) GLYCERIN (UNII: PDC6A3C0OX) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) DOCOSANOL (UNII: 9G1OE216XY) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) PROPANEDIOL (UNII: 5965N8W85T) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) CARTHAMUS TINCTORIUS (SAFFLOWER) OLEOSOMES (UNII: 9S60Q72309) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) LAVENDER OIL (UNII: ZBP1YXW0H8) LAVANDULA ANGUSTIFOLIA SUBSP. ANGUSTIFOLIA FLOWERING TOP (UNII: 9YT4B71U8P) ROSA DAMASCENA FLOWER OIL (UNII: 18920M3T13) TAGETES MINUTA FLOWER OIL (UNII: 1T0ZMU8M8B) CITRUS SINENSIS FLOWER OIL (UNII: AJ56JP5TFP) ETHYL FERULATE (UNII: 5B8915UELW) UNDARIA PINNATIFIDA (UNII: ICV1OK7M1S) POLYGLYCERYL-3 PENTARICINOLEATE (UNII: 7Q0OK5DOT4) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) BORAGE SEED OIL (UNII: F8XAG1755S) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) XYLITYLGLUCOSIDE (UNII: O0IEZ166FB) COCO GLUCOSIDE (UNII: ICS790225B) LEVOMENOL (UNII: 24WE03BX2T) XANTHAN GUM (UNII: TTV12P4NEE) ISOSTEARIC ACID (UNII: X33R8U0062) ANHYDROXYLITOL (UNII: 8XWR7NN42F) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) XYLITOL (UNII: VCQ006KQ1E) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) POLYGONUM AVICULARE TOP (UNII: ZCD6009IUF) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM BENZOATE (UNII: OJ245FE5EU) SAFFLOWER OIL (UNII: 65UEH262IS) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) TREHALOSE (UNII: B8WCK70T7I) LUTEIN (UNII: X72A60C9MT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54111-152-50 1 in 1 CARTON 01/31/2021 1 50 mL in 1 JAR; Type 0: Not a Combination Product 2 NDC:54111-152-51 1 in 1 CARTON 01/31/2021 2 15 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/31/2021 Labeler - Bentley Laboratories, LLC (068351753)