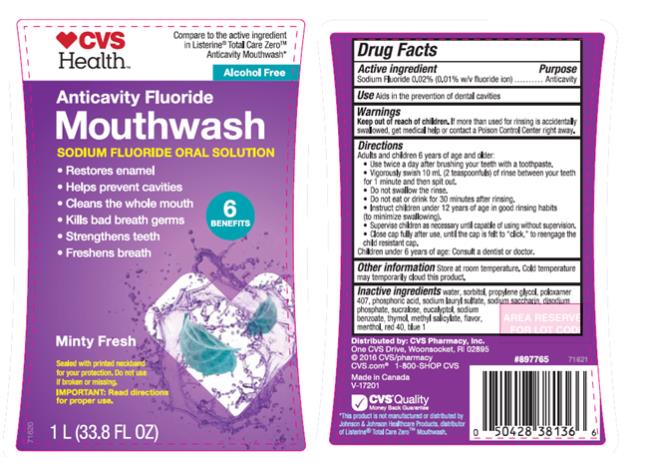
Label: ANTICAVITY FLUORIDE- sodium fluoride mouthwash
-
Contains inactivated NDC Code(s)
NDC Code(s): 69842-232-15 - Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 1, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
- Warnings
-
Directions
Adults and children 6 years of age and older:
Use twice a day after brushing your teeth with a toothpaste.
Vigorously swish 10 mL (2 teaspoonfuls) of rinse between your teeth for 1 minute and then spit out.
Do not swallow the rinse.
Do not eat or drink for 30 minutes after rinsing.
Instruct children 12 years of age in good rinsing habits (to minimize swallowing).
Supervise children as necessary until capable of using without supervision.
Close cap fully after use, until the cap is felt to “click”, to reengage the child resistant cap.
Children under 6 years of age: Consult a dentist or doctor
Other information Store at room temperature. Cold temperature may temporarily cloud this product.
-
Inactive ingredients
water, sorbitol, propylene glycol, poloxamer 407, phosphoric acid, sodium lauryl sulfate, sodium saccharin, disodium phosphate, sucralose, eucalyptol, sodium benzoate, thymol, methyl salicylate, flavor, menthol, red 40, blue 1.
This rinse may cause temporary staining to the surface of teeth.
This is not harmful and adequate brushing may prevent its occurrence.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTICAVITY FLUORIDE
sodium fluoride mouthwashProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-232 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.1 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POLOXAMER 407 (UNII: TUF2IVW3M2) PHOSPHORIC ACID (UNII: E4GA8884NN) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) SUCRALOSE (UNII: 96K6UQ3ZD4) EUCALYPTOL (UNII: RV6J6604TK) SODIUM BENZOATE (UNII: OJ245FE5EU) THYMOL (UNII: 3J50XA376E) METHYL SALICYLATE (UNII: LAV5U5022Y) MENTHOL (UNII: L7T10EIP3A) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-232-15 1000 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/30/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part355 01/30/2017 Labeler - CVS Pharmacy (062312574)