Label: PIPETTE ECZEMA- oatmeal liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 73517-001-01, 73517-001-06 - Packager: Amyris, Inc
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 6, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
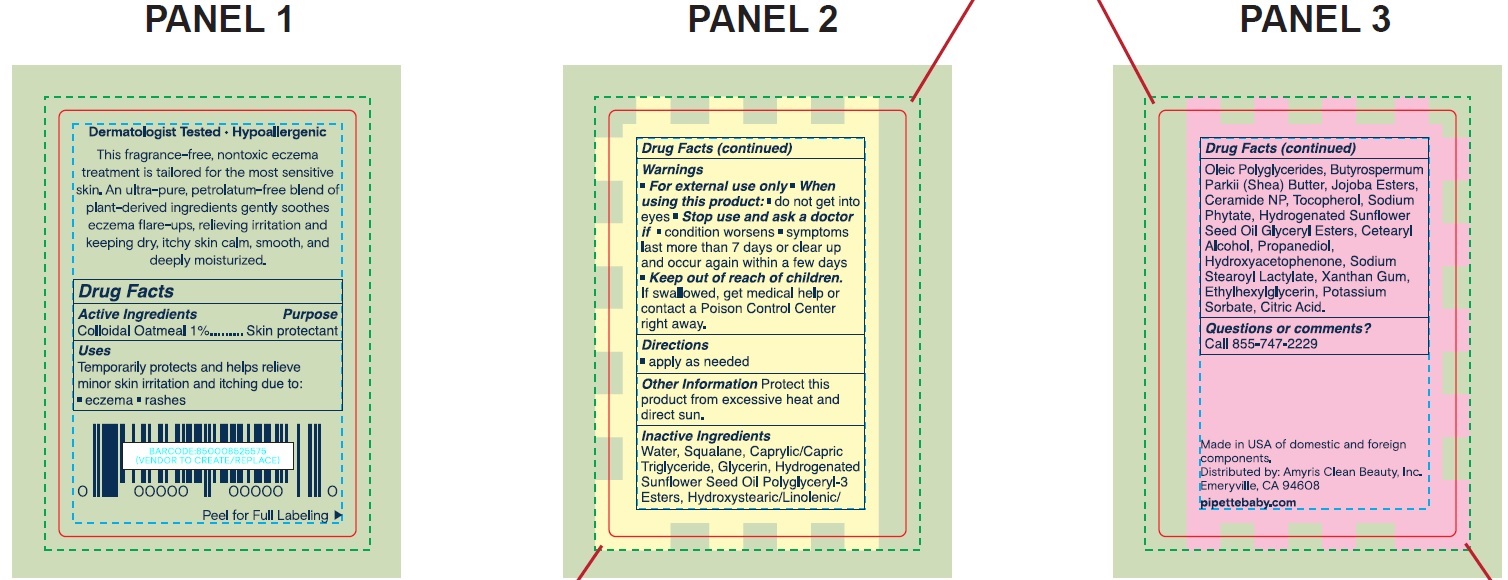
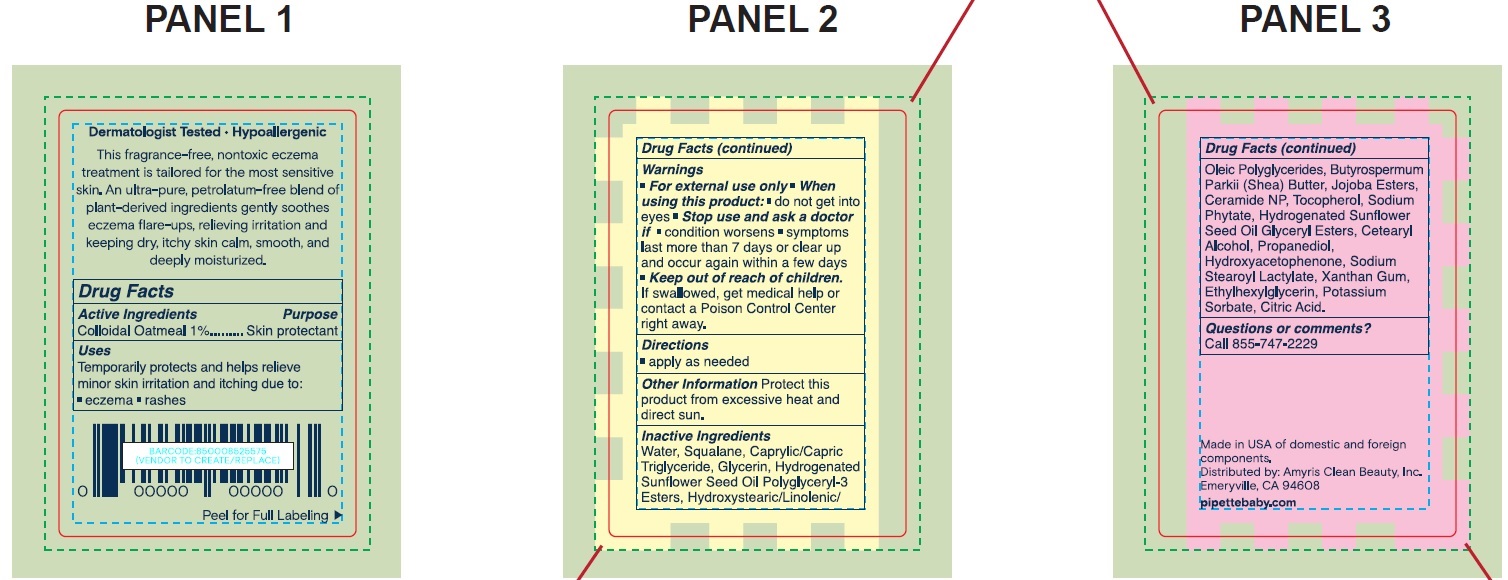
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Other Information
-
Inactive Ingredients
Water, Squalane, Caprylic/Capric Triglyceride, Glycerin, Hydrogenated Sunflower Seed Oil Polyglyceryl-3 Esters, Hydroxystearic/Linolenic/Oleic Polyglycerides, Butyrospermum Parkii (Shea) Butter, Jojoba Esters, Ceramide NP, Tocopherol, Sodium Phytate, Hydrogenated Sunflower Seed Oil, Glyceryl Esters, Cetearyl Alcohol, Propanediol, Hydroxyacetophenone, Sodium Stearoyl Lactylate, Xanthan Gum, Ethylhexylglycerin, Potassium Sorbate, Citric Acid.
- Questions or comments?
- Package Labeling:73517-001-01
- Package Labeling:73517-001-06
-
INGREDIENTS AND APPEARANCE
PIPETTE ECZEMA
oatmeal liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73517-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OATMEAL (UNII: 8PI54V663Y) (OATMEAL - UNII:8PI54V663Y) OATMEAL 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SQUALANE (UNII: GW89575KF9) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) GLYCERIN (UNII: PDC6A3C0OX) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE NP (UNII: 4370DF050B) TOCOPHEROL (UNII: R0ZB2556P8) PHYTATE SODIUM (UNII: 88496G1ERL) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PROPANEDIOL (UNII: 5965N8W85T) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) SODIUM STEAROYL LACTYLATE (UNII: IN99IT31LN) XANTHAN GUM (UNII: TTV12P4NEE) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73517-001-01 1 in 1 BOX 05/16/2022 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:73517-001-06 1 in 1 BOX 05/16/2022 2 177 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 05/16/2022 Labeler - Amyris, Inc (185930182)