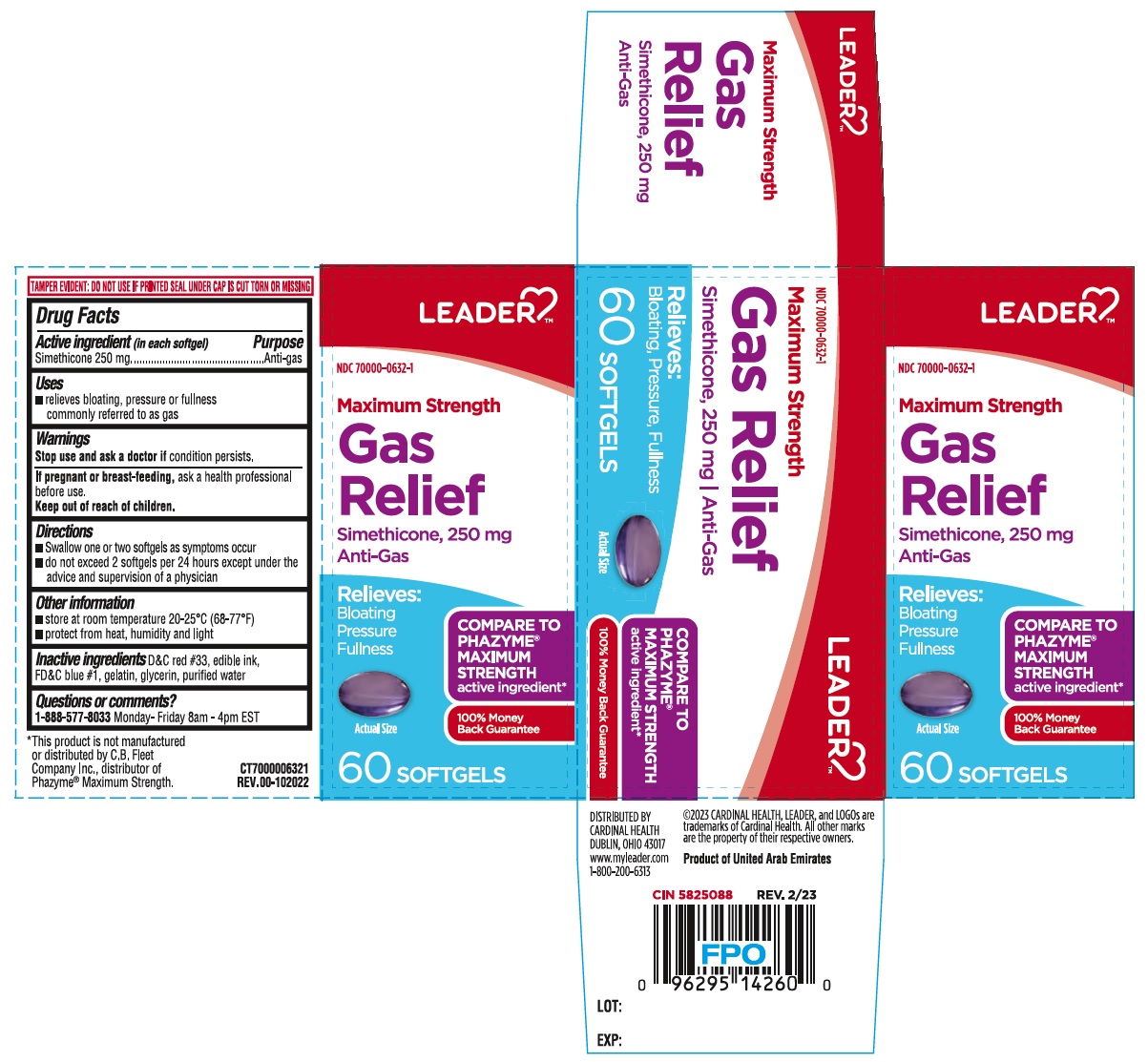
Label: MAXIMUM STRENGTH GAS RELIEF SOFTGEL- simethicone capsule, liquid filled
- NDC Code(s): 70000-0632-1
- Packager: Cardinal Health 110, LLC. DBA Leader
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 11, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient (in each softgel)
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
LEADER™
Relieves:
Bloating
Pressure
FullnessCOMPARE TO PHAZYME® MAXIMUM STRENGTH active ingredient*
100% Money Back Guarantee
TAMPER EVIDENT: DO NOT USE IF PRINTED SEAL UNDER CAP IS CUT TORN OR MISSING
*This product is not manufactured or distributed by C.B. Fleet Company Inc., distributor of Phazyme® Maximum Strength.
DISTRIBUTED BY
CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com
1-800-200-6313©2023 CARDINAL HEALTH, LEADER, and LOGOs are trademarks of Cardinal Health. All other marks are the property of their respective owners.
Product of United Arab Emirates
- Packaging
-
INGREDIENTS AND APPEARANCE
MAXIMUM STRENGTH GAS RELIEF SOFTGEL
simethicone capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0632 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 250 mg Inactive Ingredients Ingredient Name Strength D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Product Characteristics Color purple Score no score Shape OVAL Size 12mm Flavor Imprint Code 813 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0632-1 1 in 1 CARTON 02/20/2023 1 60 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M002 02/20/2023 Labeler - Cardinal Health 110, LLC. DBA Leader (063997360)