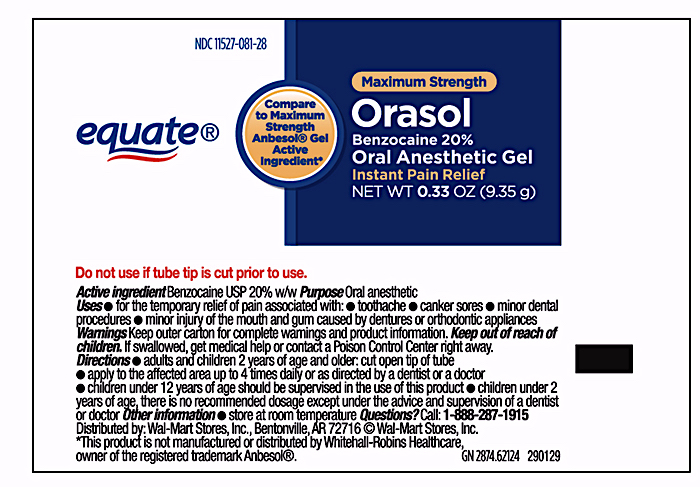
Label: EQUATE ORAL PAIN RELIEF- benzocaine gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 11527-081-28 - Packager: Sheffield Pharmaceuticals LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 18, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
- Warnings
-
Directions
- do not use tube if it is cut prior to opening
- cut open tip of tube on score mark
- use your fingertip or cotton applicator to apply a small pea-size amount of Oral Pain Relief Gel
- apply to affected area up to four times daily or as directed by a dentist or physician
- Adults and children 2 years of age and older: Apply to affected area
- Children under 12 years of age should be supervised in the use of this product
- Children under 2 years of age: Consult a doctor
- Other information
- Inactive ingredients
- Principal Display Panel – 0.33oz Carton Label
- Principal Display Panel – 0.33oz Tube Label
-
INGREDIENTS AND APPEARANCE
EQUATE ORAL PAIN RELIEF
benzocaine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11527-081 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg in 1 g Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL 300 (UNII: 5655G9Y8AQ) BENZYL ALCOHOL (UNII: LKG8494WBH) CARBOMER 934 (UNII: Z135WT9208) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Saccharin Sodium (UNII: SB8ZUX40TY) METHYLPARABEN (UNII: A2I8C7HI9T) GLYCERIN (UNII: PDC6A3C0OX) Product Characteristics Color red Score Shape Size Flavor SPEARMINT (SPEARMINT) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11527-081-28 1 in 1 CARTON 01/23/2007 12/31/2023 1 9.35 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 01/23/2007 Labeler - Sheffield Pharmaceuticals LLC (151177797) Establishment Name Address ID/FEI Business Operations Sheffield Pharmaceuticals LLC 151177797 MANUFACTURE(11527-081)