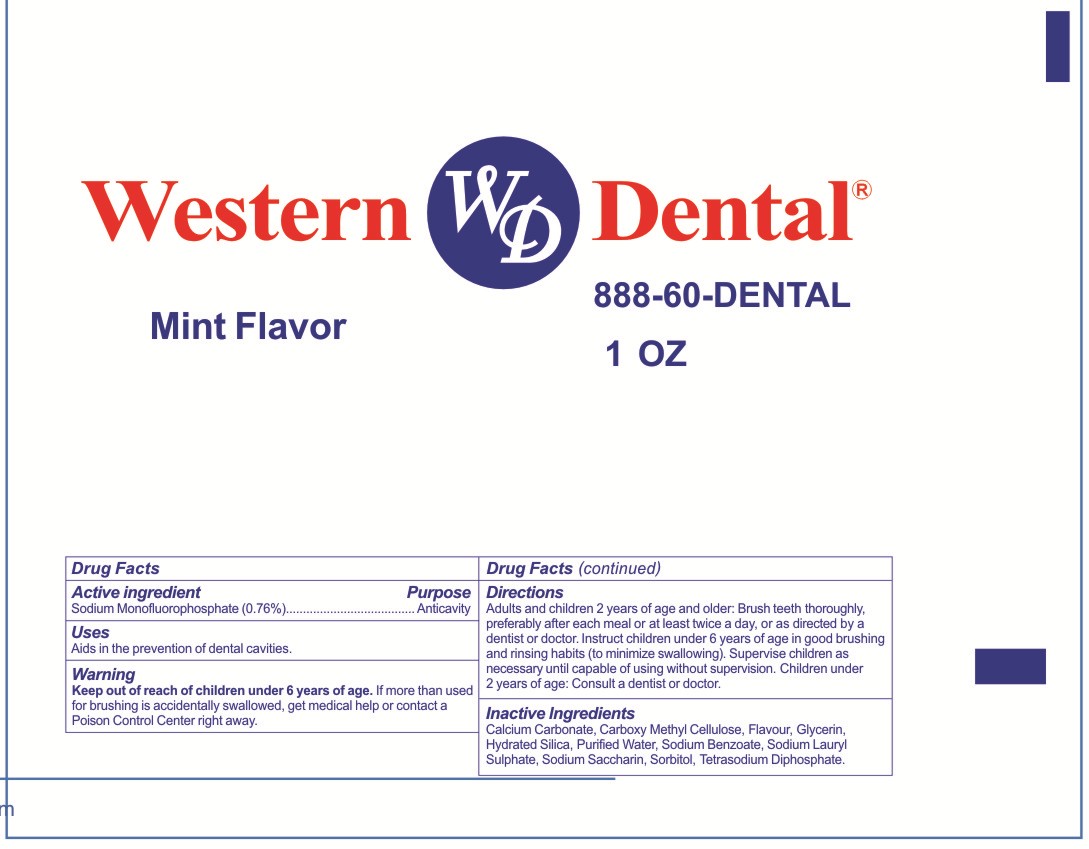
Label: WESTERN DENTAL- sodium fluoride paste
-
Contains inactivated NDC Code(s)
NDC Code(s): 71035-800-01 - Packager: Yangzhou Hongshengding Chemical Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 19, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Uses
- Warnings
-
Directions
Adults and children 2years of age and older:Brush teeth thoroughly,preferably after each meal or at least twice a day,or as directed by a dentist or a doctor.
Instruct children under 6 years of age in good brushing and rinsing habits (to minimize swallowing),
supervise children as necessary until capable of useing without supervision.Children under 2 years of age:consult a dentist or doctor.
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
WESTERN DENTAL
sodium fluoride pasteProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71035-800 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM MONOFLUOROPHOSPHATE (UNII: C810JCZ56Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.76 g in 100 g Inactive Ingredients Ingredient Name Strength SODIUM LAURYL SULFATE (UNII: 368GB5141J) CALCIUM CARBONATE (UNII: H0G9379FGK) CARBOXYMETHYLCELLULOSE (UNII: 05JZI7B19X) GLYCERIN (UNII: PDC6A3C0OX) SACCHARIN SODIUM (UNII: SB8ZUX40TY) WATER (UNII: 059QF0KO0R) HYDRATED SILICA (UNII: Y6O7T4G8P9) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) PHYTONADIOL TETRASODIUM DIPHOSPHATE (UNII: X952T8E4RN) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71035-800-01 6 in 1 CARTON 02/11/2016 1 24 in 1 BOX 1 28.35 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 02/11/2016 Labeler - Yangzhou Hongshengding Chemical Co.,Ltd. (543774755) Registrant - Yangzhou Hongshengding Chemical Co.,Ltd. (543774755) Establishment Name Address ID/FEI Business Operations Yangzhou Hongshengding Chemical Co.,Ltd. 543774755 manufacture(71035-800)