Label: ANTI ITCH- diphenhydramine hydrochloride, zinc acetate cream
- NDC Code(s): 41250-622-16, 41250-622-64
- Packager: Meijer Distribution Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 9, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
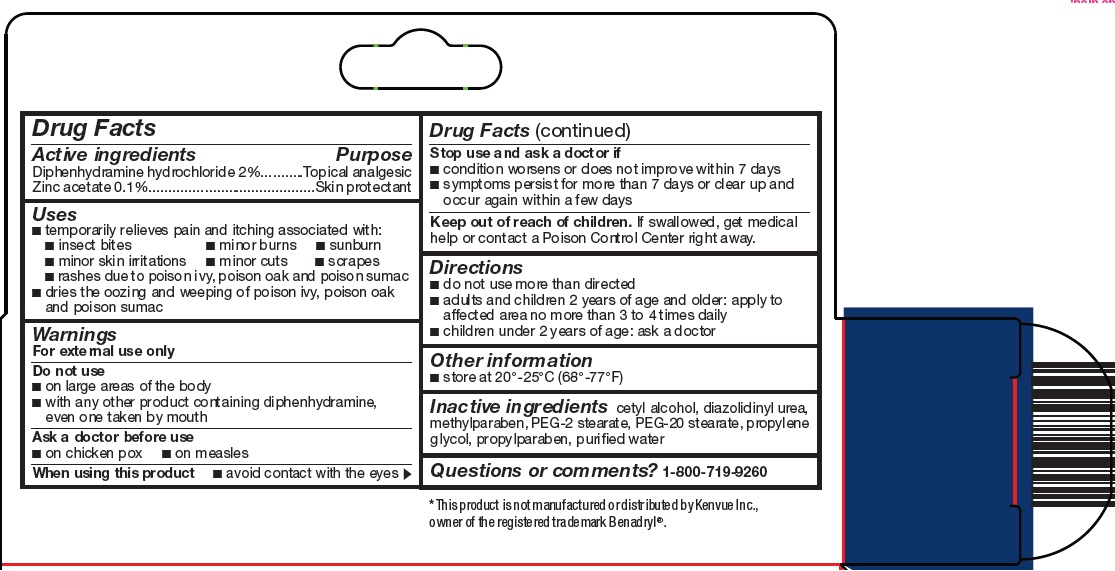
- Active ingredients
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- •
- on large areas of the body
- •
- with any other product containing diphenhydramine, even one taken by mouth
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
meijer®
EXTRA STRENGTH
Anti-Itch Cream
TOPICAL ANALGESIC | SKIN PROTECTANT
Itch Stopping Cream
Insect Bites | Poison Ivy, Oak, Sumac
Mosquito Bites | Sunburn | Minor Cuts & Scrapes
Fast Relief for Most Outdoor Itches
meijer®
EXTRA STRENGTH
Anti-Itch Cream
COMPARE TO EXTRA STRENGTH BENADRYL® ITCH STOPPING CREAM ACTIVE INGREDIENTS
TOPICAL ANALGESIC | SKIN PROTECTANT
Itch And Rash Relief
NET WT 1 OZ (28 g)
-
INGREDIENTS AND APPEARANCE
ANTI ITCH
diphenhydramine hydrochloride, zinc acetate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41250-622 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 2 g in 100 g ZINC ACETATE (UNII: FM5526K07A) (ZINC CATION - UNII:13S1S8SF37) ZINC ACETATE 0.1 g in 100 g Inactive Ingredients Ingredient Name Strength CETYL ALCOHOL (UNII: 936JST6JCN) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) DIETHYLENE GLYCOL MONO- AND DIPALMITOSTEARATE (UNII: 94YQ11Y95F) PEG-20 STEARATE (UNII: NBX892EA57) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41250-622-16 2 in 1 CARTON 04/20/2010 06/01/2018 1 28 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:41250-622-64 1 in 1 CARTON 03/17/2010 2 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 03/17/2010 Labeler - Meijer Distribution Inc (006959555)