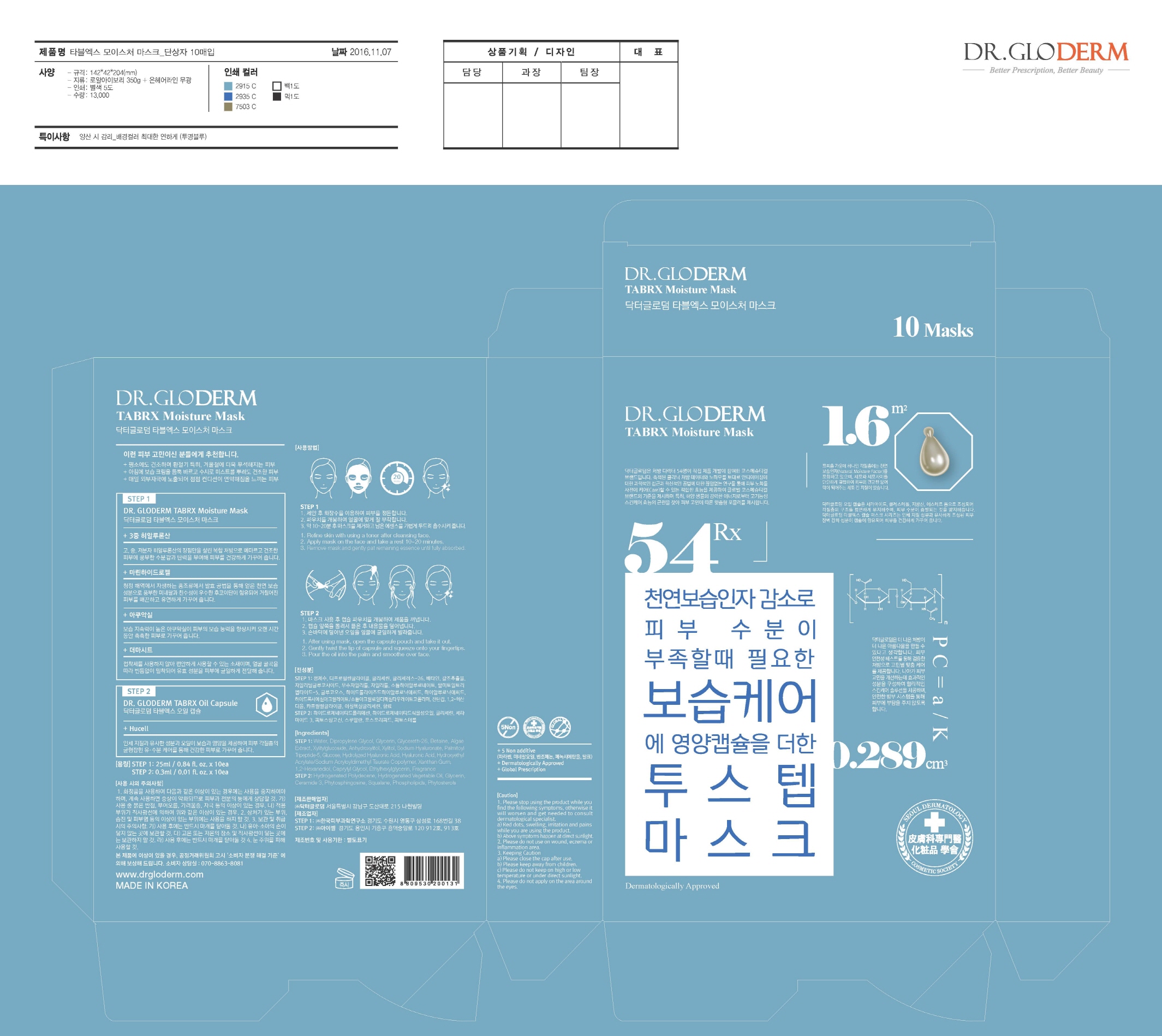
Label: DR. GLODERM TABRX MOISTURE MASK- glycerin kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 71342-0005-1 - Packager: DR.GLODERM
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 31, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
1. If the following symptoms occur after product use, stop using the product immediately and consult a dermatologist (continuous use can exacerbate the symptoms).
1) Occurrence of red spots, swelling, itchiness, and other skin irritation
2) If the symptoms above occur after the application area is exposed to direct sunlight2. Do not use on open wounds, eczema, and other skin irritations
3. Precaution for Storage and Handling
1) Close the lid after use
2) Keep out of reach of infants and children
3) Do not to store in a place with high/low temperature and exposed to direct sunlight4. Use as avoiding eye areas.
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Step1
- Refine skin with using a toner after cleansing face.
- Apply mask on the face and take a rest 10~20 minutes.
- Remove mask and gently pat remaining essence until fully absorbed.
Step2
- After using mask, open the capsule pouch and take it out.
- Gently twist the tip of capsule and squeeze onto your fingertips.
- Pour the oil into the palm and smoothe over face.
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DR. GLODERM TABRX MOISTURE MASK
glycerin kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71342-0005 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71342-0005-1 10 in 1 PACKAGE 03/27/2017 1 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 POUCH 25 mL Part 2 1 POUCH 0.3 mL Part 1 of 2 DR. GLODERM TABRX MOISTURE MASK
glycerin liquidProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 5 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 25 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other Part 2 of 2 DR. GLODERM TABRX OILCAPSULE
glycerin oilProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 1 g in 100 mL Inactive Ingredients Ingredient Name Strength SQUALANE (UNII: GW89575KF9) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.3 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part347 03/27/2017 Labeler - DR.GLODERM (694773267) Registrant - DR.GLODERM (694773267) Establishment Name Address ID/FEI Business Operations EYESEL Co., Ltd. 557836898 manufacture(71342-0005) Establishment Name Address ID/FEI Business Operations KOREA DERMAL RESEARCH CENTER CO., LTD. 694640098 manufacture(71342-0005) Establishment Name Address ID/FEI Business Operations DR.GLODERM 694773267 label(71342-0005)