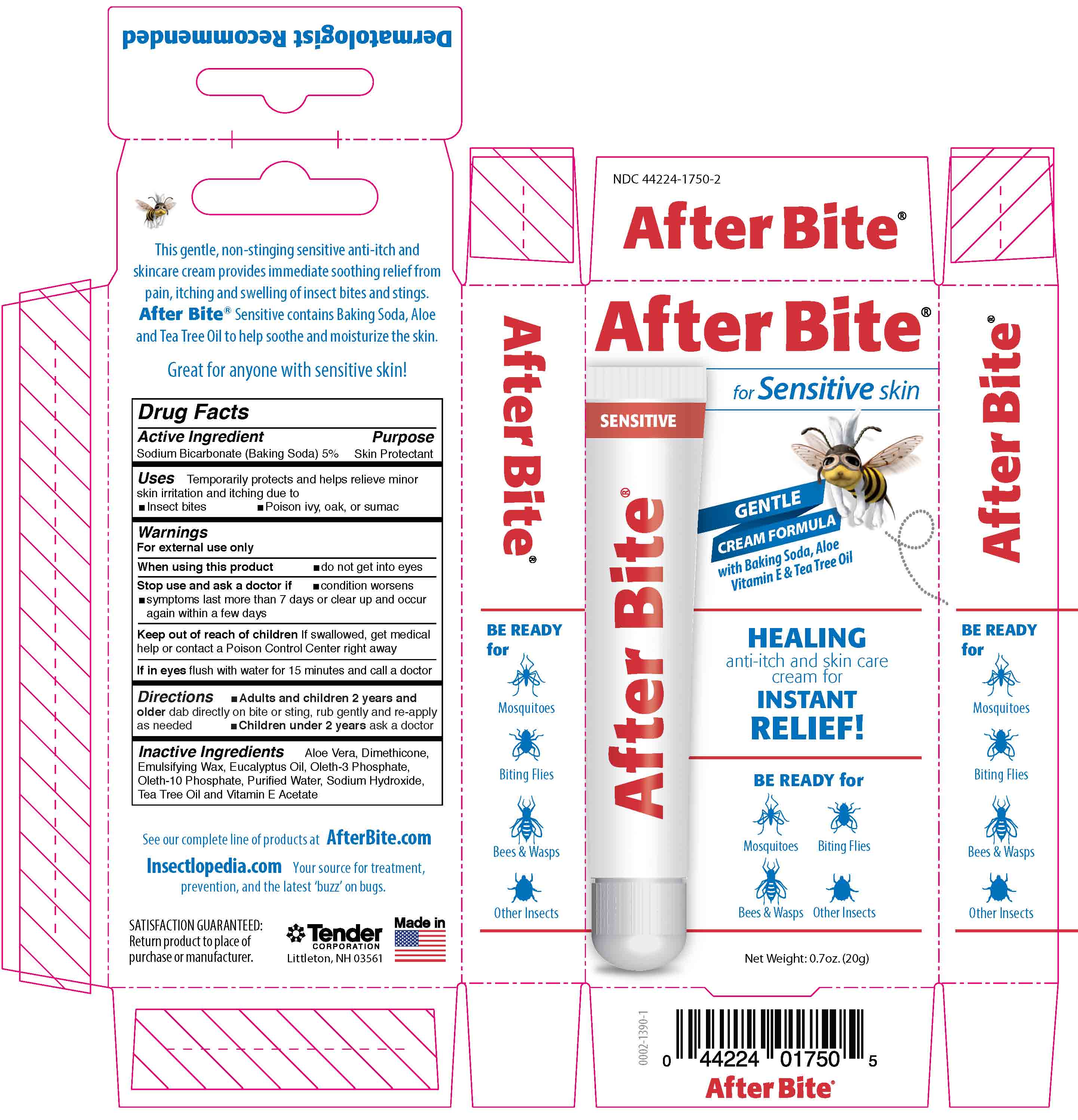
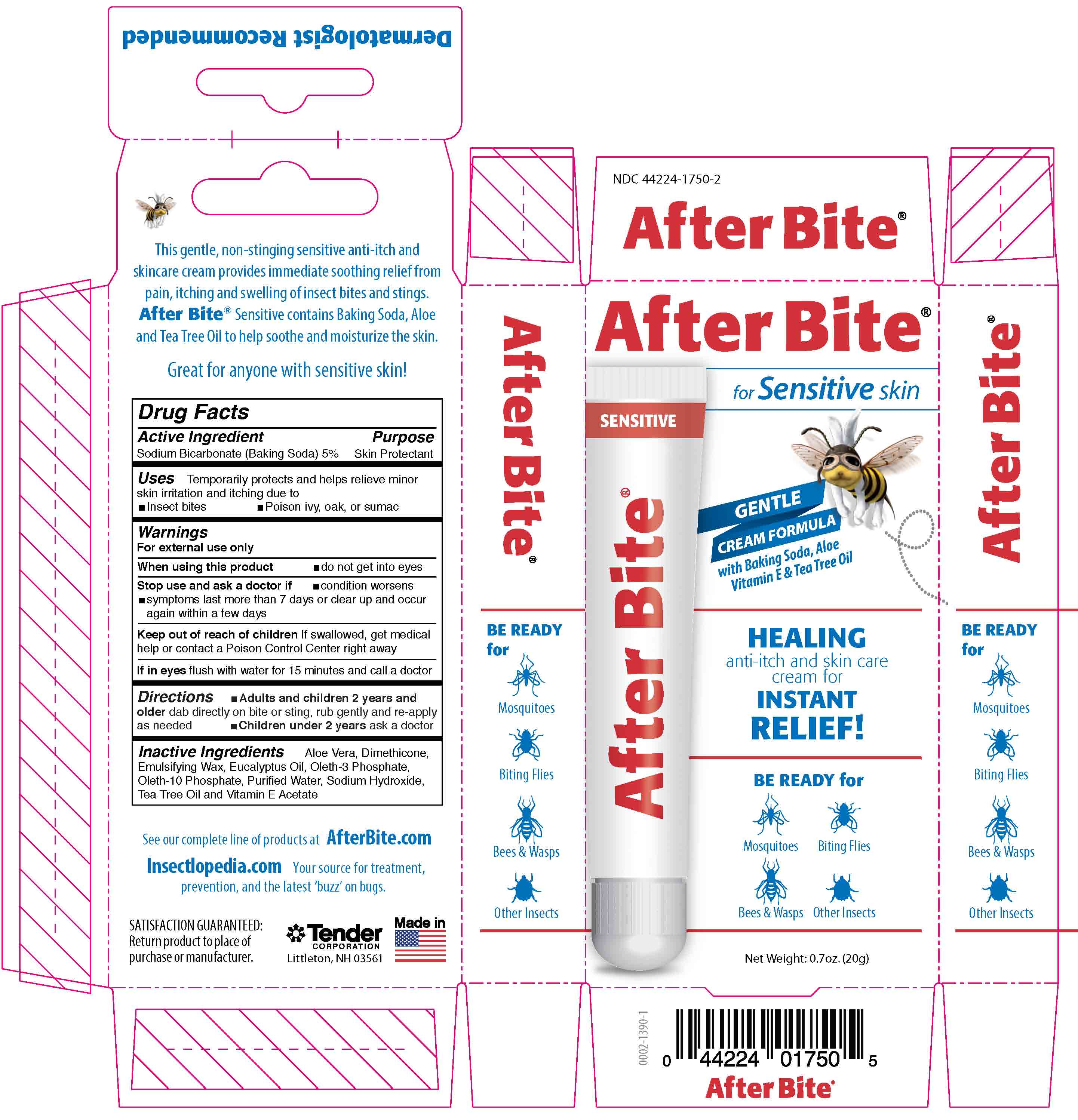
Label: AFTER BITE SENSITIVE- sodium bicarbonate liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 44224-1750-0, 44224-1750-2 - Packager: Tender Corporation
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 15, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Purpose
- Uses
- Warnings
- Directions
- Inactive Ingredients
-
After Bite Sensitive
After Bite Sensitive
for Sensitive skin
Gentle cream formula with Baking Soda, Aloe, Vitamin E & Tea Tree Oil
Healking anti-itch and skin care cream for Instant Relief!
Be Ready For: Mosquitoes, Biting Flies, Bees & Wasps and Other Insects
This gentle, non-stinging sensitive anti-itch and skincare cream provides immediate soothing relief from pain, itching and swellling of insect bites and stings. After Bite Sensititve contains Baking Soda, Aloe and Tea Tree Oil to help soothe and moisturize the Skin.
Great for anyone with Sensitive Skin!
-
INGREDIENTS AND APPEARANCE
AFTER BITE SENSITIVE
sodium bicarbonate liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:44224-1750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM BICARBONATE 5 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) DIMETHICONE (UNII: 92RU3N3Y1O) EUCALYPTUS OIL (UNII: 2R04ONI662) OLETH-3 PHOSPHATE (UNII: 8Q0Z18J1VL) OLETH-10 PHOSPHATE (UNII: Q95361F4VS) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) TEA TREE OIL (UNII: VIF565UC2G) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:44224-1750-2 1 in 1 BOX 11/26/2014 1 NDC:44224-1750-0 20 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 11/26/2014 Labeler - Tender Corporation (064437304) Establishment Name Address ID/FEI Business Operations Tender Corporation 064437304 manufacture(44224-1750)