Label: CVS HEALTH SPF 100 ULTRA SHEER- avobenzone, homosalate, octisalate, octocrylene, oxybenzone lotion
- NDC Code(s): 69842-125-09
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 19, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
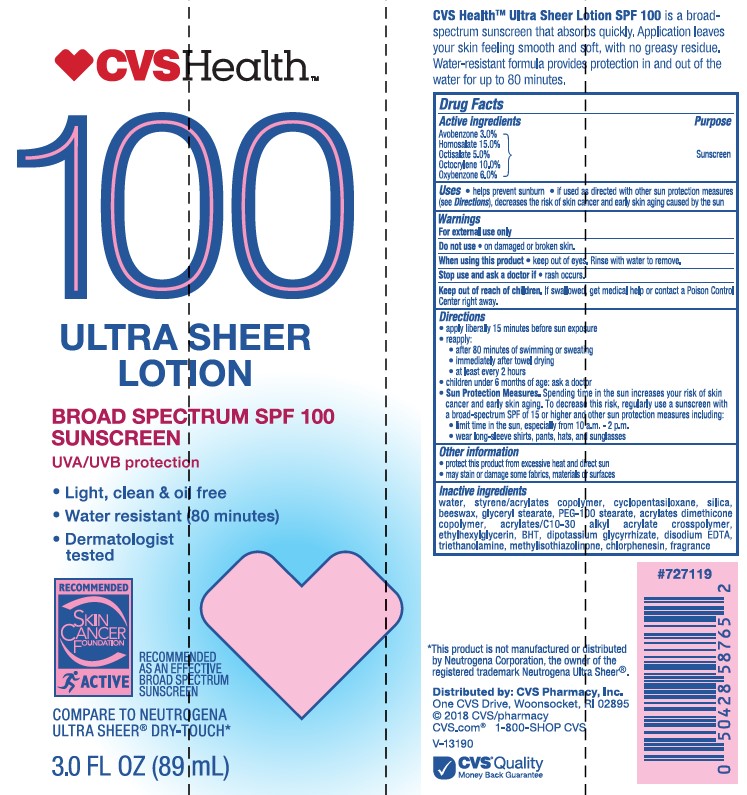
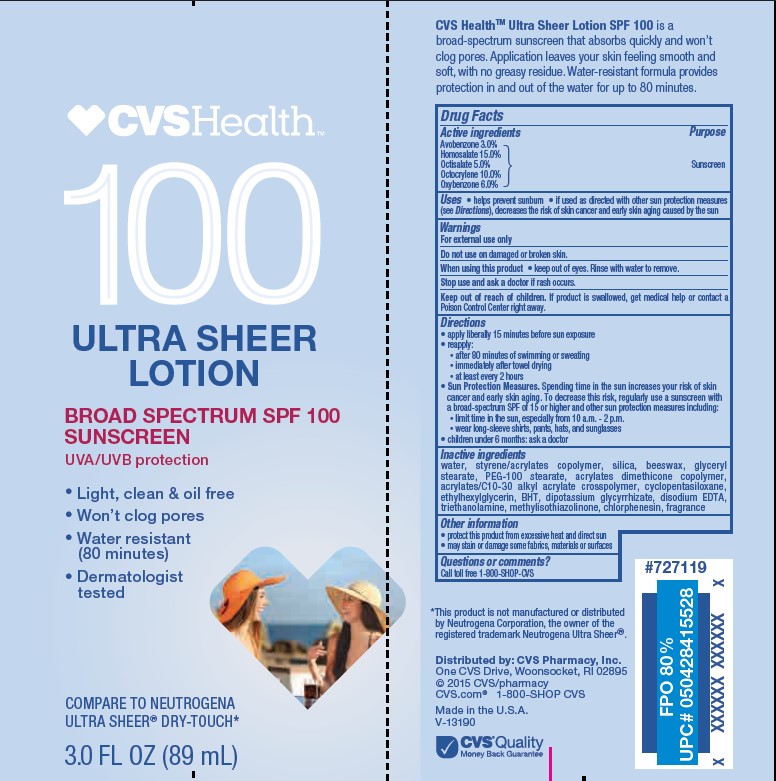
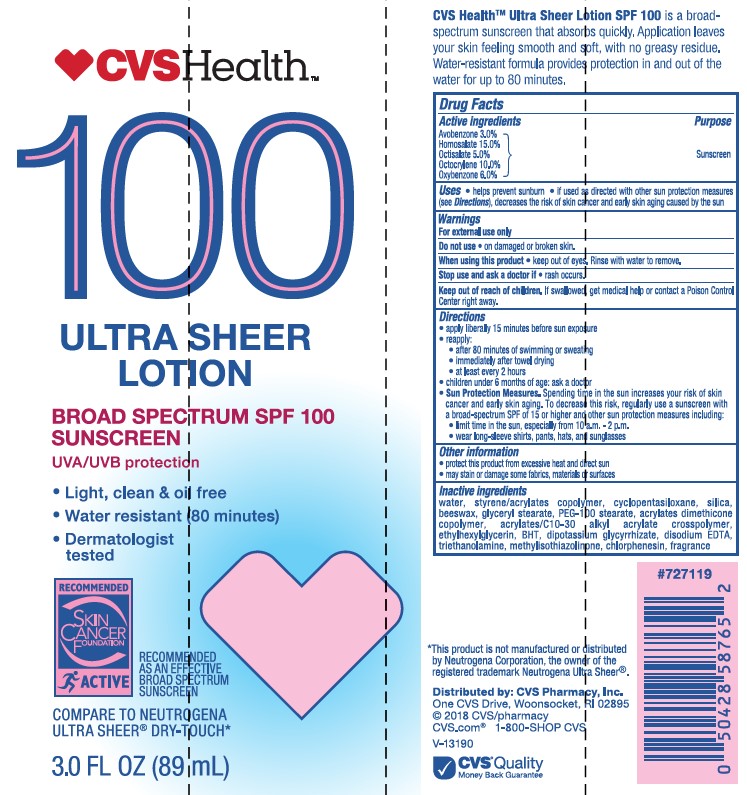
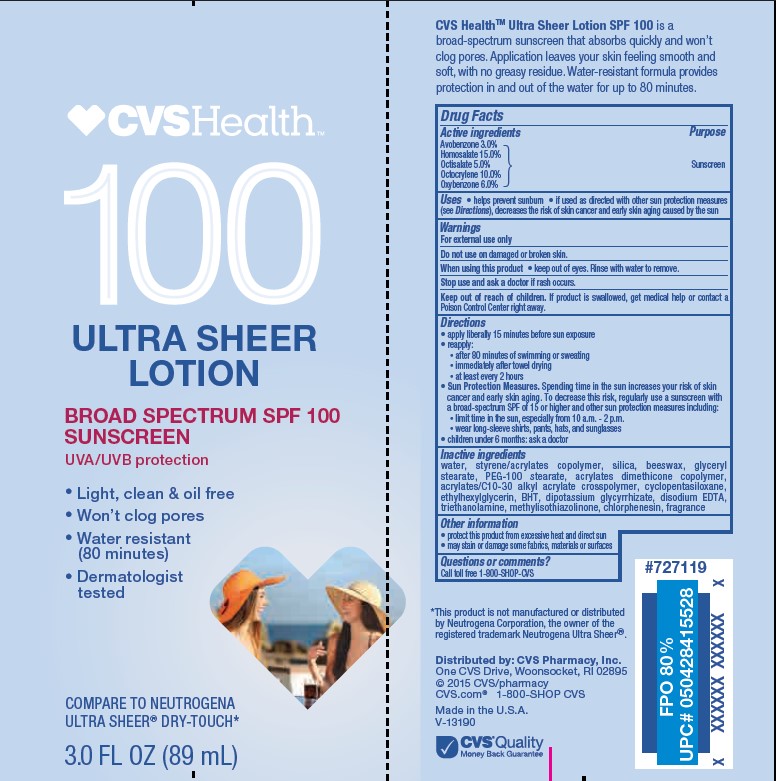
- Active ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor
- Keep out of reach of children.
-
Directions
• apply liberally 15 minutes before sun exposure
• reapply:
• after 80 minutes of swimming or sweating
• immediately after towel drying
• at least every 2 hours• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
• children under 6 months: Ask a doctor -
Inactive ingredients
water, styrene/acrylates copolymer, silica, beeswax, glyceryl stearate, PEG-100 stearate, acrylates dimethicone copolymer, acrylates/C10-30 alkyl acrylate crosspolymer, cyclopentasiloxane, ethylhexylglycerin, BHT, dipotassium glycyrrhizate, disodium EDTA, triethanolamine, methylisothiazolinone, chlorphenesin, fragrance
- Other information
- Label
-
INGREDIENTS AND APPEARANCE
CVS HEALTH SPF 100 ULTRA SHEER
avobenzone, homosalate, octisalate, octocrylene, oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-125 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 150 mg in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 60 mg in 1 mL Inactive Ingredients Ingredient Name Strength BUTYL METHACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID/STYRENE CROSSPOLYMER (UNII: V5RS026Q0H) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) TROLAMINE (UNII: 9O3K93S3TK) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) WATER (UNII: 059QF0KO0R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) YELLOW WAX (UNII: 2ZA36H0S2V) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) CHLORPHENESIN (UNII: I670DAL4SZ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-125-09 89 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/23/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 10/23/2015 Labeler - CVS Pharmacy (062312574)