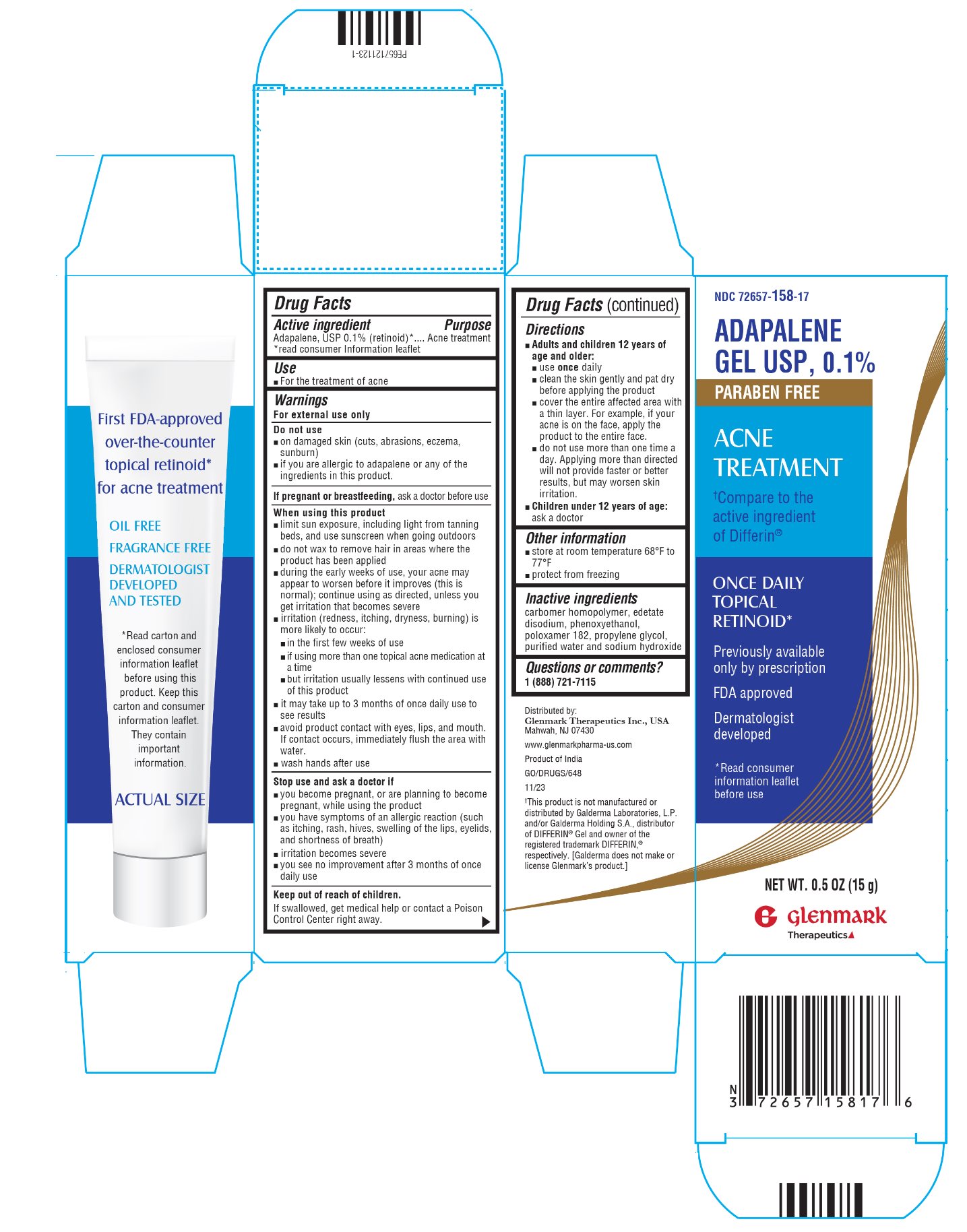
Label: ADAPALENE gel
- NDC Code(s): 72657-158-17, 72657-158-55
- Packager: GLENMARK THERAPEUTICS INC., USA
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 25, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Use
-
Warnings
For external use only
Do not use
- •
- on damaged skin (cuts, abrasions, eczema, sunburn)
- •
- if you are allergic to adapalene or any of the ingredients in this product
If pregnant or breastfeeding, ask a doctor before use
When using this product
- •
- limit sun exposure, including light from tanning beds, and use sunscreen when going outdoors
- •
- do not wax to remove hair in areas where the product has been applied
- •
- during the early weeks of use, your acne may appear to worsen before it improves (this is normal); continue using as directed, unless you get irritation that becomes severe
- •
- irritation (redness, itching, dryness, burning) is more likely to occur:
- •
- in the first few weeks of use
- •
- if using more than one topical acne medication at a time
- •
- but irritation usually lessens with continued use of this product
- •
- it may take up to 3 months of once daily use to see results
- •
- avoid product contact with eyes, lips, and mouth. If contact occurs, immediately flush the area with water.
- •
- wash hands after use
Stop use and ask doctor if
- •
- you become pregnant, or are planning to become pregnant, while using the product
- •
- you have symptoms of an allergic reaction (such as itching, rash, hives, swelling of the lips, eyelids, and shortness of breath)
- •
- irritation becomes severe
- •
- you see no improvement after 3 months of once daily use
- Keep out of reach of children.
-
Directions
- •
- Adults and children 12 years of age and older:
- •
- use once daily
- •
- clean the skin gently and pat dry before applying the product
- •
- cover the entire affected area with a thin layer. For example, if your acne is on the face, apply the product to the entire face.
- •
- do not use more than one time a day. Applying more than directed will not provide faster or better results, but may worsen skin irritation.
- •
- Children under 12 years of age: ask a doctor
- Other information
- Inactive Ingredients
- Questions or comments?
-
Patient Leaflet
ADAPALENE GEL USP, 0.1%
ACNE TREATMENT
Frequently Asked Questions
What is adapalene gel 0.1% and what is it used for?
- •
- Adapalene gel 0.1% is a topical retinoid medication used for the treatment of acne in people age 12 years and older.
- •
- Use only one time a day.
What should I know before using the product?
- •
- If pregnant or breastfeeding, ask a doctor before use.
- •
- Some other retinoid drugs have been shown to cause birth defects. There is no specific evidence that adapalene gel 0.1% causes birth defects in humans when used topically as directed.
How often do I apply the product?
- •
- Apply this product only one time a day and try to apply the product at the same time each day.
How do I apply the product?
- •
- Gently clean the affected areas using a mild (non-irritating) cleanser and pat dry.
- •
- Apply adapalene gel 0.1% as a thin layer to the affected areas of the skin only one time a day. For example, if you get acne on the face, clean, dry and apply the product to the entire face. Adapalene gel 0.1% is not a spot treatment and should not be used to treat a single pimple.
How long will it take for adapalene gel 0.1% to work?
- •
- It may take up to 3 months of daily use for results to appear.
- •
- Do not use more than one time a day. Applying more than directed will not provide faster or better results, but may worsen skin irritation.
What do I do if I need to be in the sun?
- •
- When possible, limit sun exposure, including light from tanning beds.
- •
- When going outdoors, use a sunscreen as labeled. Your skin may be more sensitive while using adapalene gel 0.1%. If you use this product during the day, allow it to dry before applying sunscreen.
Can I use a moisturizer if my skin is dry?
- •
- Yes, use of a moisturizer may help decrease dryness and other signs of irritation. Avoid products containing alpha hydroxy or glycolic acids which may worsen irritation.
When is my skin most likely to become irritated? And what do I do?
- •
- Irritation (redness, itching, dryness, burning) is more likely to occur:
- •
- In the first few weeks of use.
What do I do if my skin becomes severely irritated?
- •
- If irritation becomes severe (overly bothersome), stop use and ask a doctor before using the product again.
Can I remove unwanted facial hair by waxing while using this product?
- •
- Do not use wax to remove hair in areas where the product has been applied because it may worsen skin irritation.
What ingredients are used in adapalene gel 0.1%?
- •
- Adapalene gel 0.1% contains:
The active ingredient is adapalene 0.1%. The other ingredients are: carbomer homopolymer, edetate disodium, phenoxyethanol, poloxamer 182, propylene glycol, purified water and sodium hydroxide.
How should I store this product?
- •
- Adapalene gel 0.1% should be stored at room temperature [68° to 77°F]. Protect from freezing.
- •
- Do not use the product after the expiry date marked on the crimp of the tube.
Other Questions? Where can I get more information?
Phone: 1 (888) 721-7115
March 2023
- Package/Label Principal Display Panel
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ADAPALENE
adapalene gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72657-158 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADAPALENE (UNII: 1L4806J2QF) (ADAPALENE - UNII:1L4806J2QF) ADAPALENE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) EDETATE DISODIUM (UNII: 7FLD91C86K) POLOXAMER 182 (UNII: JX0HIX6OAG) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72657-158-17 1 in 1 CARTON 03/10/2023 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:72657-158-55 1 in 1 CARTON 03/10/2023 2 45 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091314 03/10/2023 Labeler - GLENMARK THERAPEUTICS INC., USA (969085666) Establishment Name Address ID/FEI Business Operations Glenmark Pharmaceuticals Limited 677318665 MANUFACTURE(72657-158) , ANALYSIS(72657-158)