Label: UNIVERSAL CHARCOAL WITH ALOE VERA ANTI-CAVITY FLUORIDE- sodium fluoride paste
- NDC Code(s): 52000-110-01
- Packager: Universal Distribution Center LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 16, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
- adults and children 12 years of age and older
- apply atleast 1-inch strip of product onto soft bristle toothbrush
- brush teeth thoroughly for atleast 1 minute twice a day (morning and evening), and not more then 3 times a day, or as recommended by a dentist or doctor.
- Make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing.
Children under 12 years of age: Consult a dentist or doctor
- SPL UNCLASSIFIED SECTION
- INACTIVE INGREDIENT
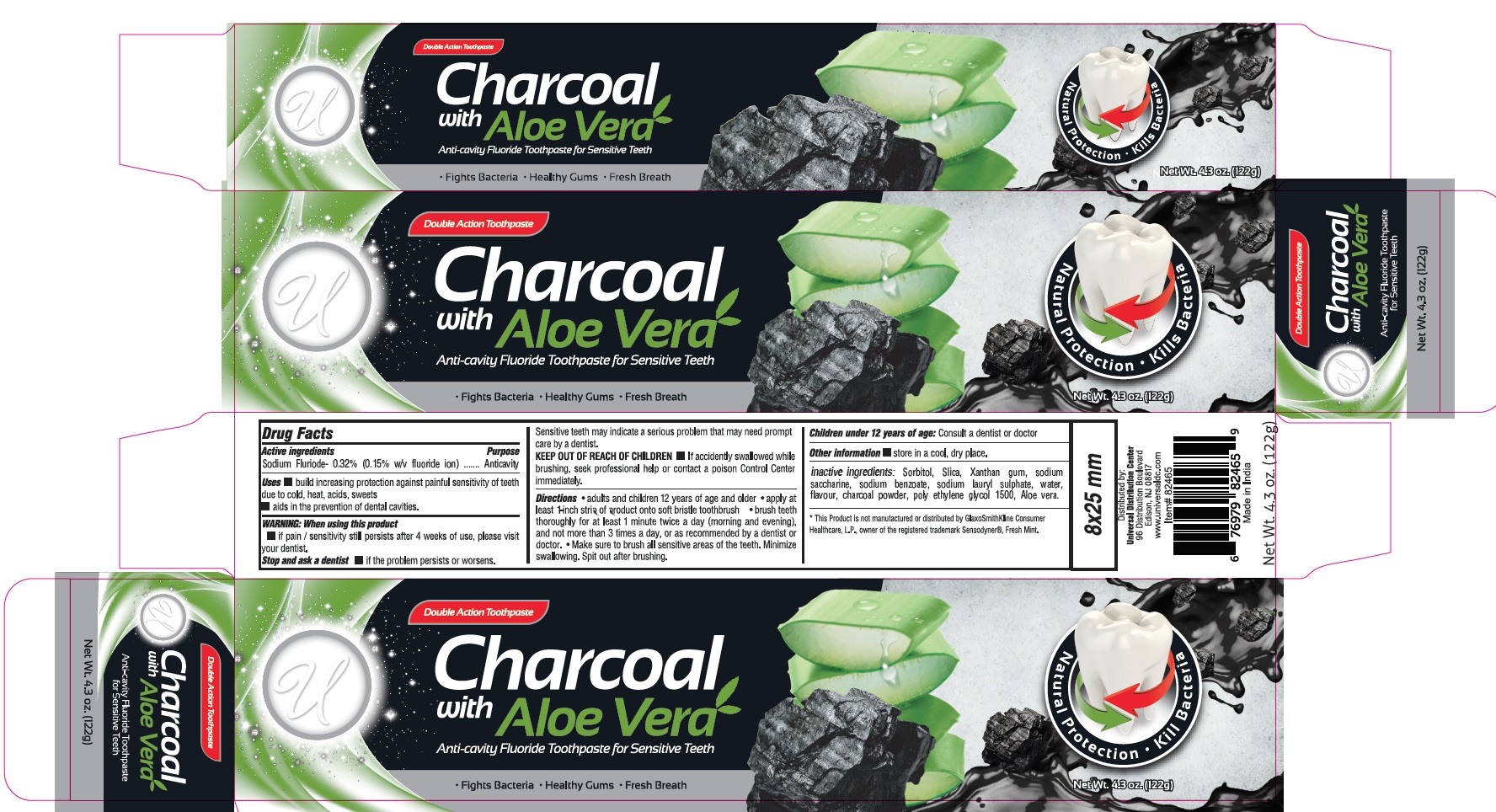
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
UNIVERSAL CHARCOAL WITH ALOE VERA ANTI-CAVITY FLUORIDE
sodium fluoride pasteProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52000-110 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.15 g in 100 g Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) XANTHAN GUM (UNII: TTV12P4NEE) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) WATER (UNII: 059QF0KO0R) ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) POLYETHYLENE GLYCOL 1600 (UNII: 1212Z7S33A) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52000-110-01 122 g in 1 TUBE; Type 0: Not a Combination Product 05/25/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 05/25/2020 Labeler - Universal Distribution Center LLC (019180459)