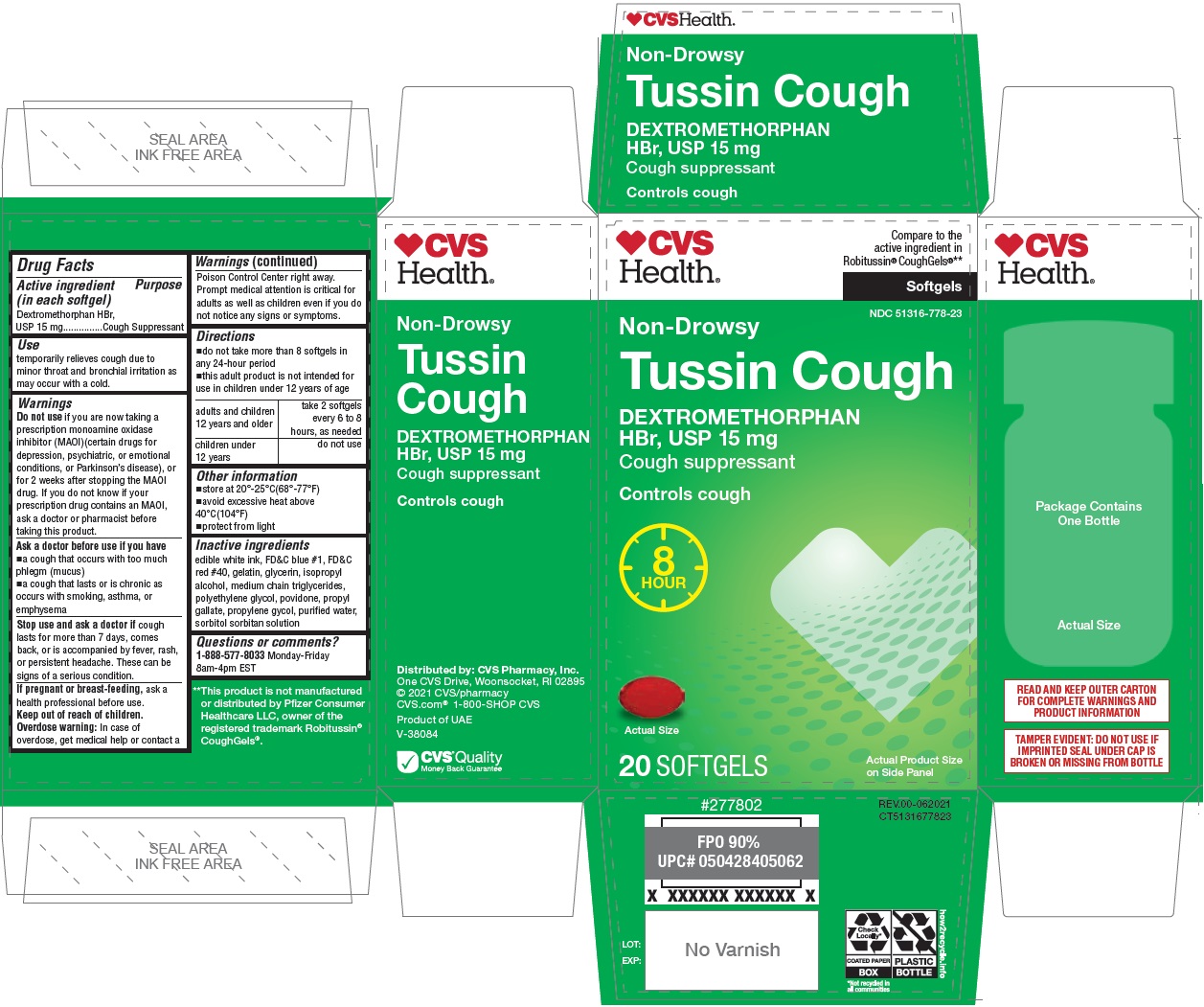
Label: LONG ACTING COUGH SOFTGELS- dextromethorphan hydrobromide capsule, liquid filled
- NDC Code(s): 51316-778-23
- Packager: CVS PHARMACY, INC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 22, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient (in each softgel)
- Purpose
- Use
-
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- a cough that occurs with too much phlegm (mucus)
- a cough that lasts or is chronic as occurs with smoking, asthma, or emphysema
Stop use and ask a doctor if cough lasts for more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. These can be signs of a serious condition
If pregnant or breast-feeding, ask a health professional before use.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
CVS Health®
Compare to the active ingredient in Robitussin® CoughGels®**
Non-Drowsy
Controls cough
8 HOUR
READ AND KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL UNDER CAP IS BROKEN OR MISSING FROM BOTTLE
**This product is not manufactured or distributed by Pfizer Consumer Healthcare LLC, owner of the registered trademark Robitussin® CoughGels®.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2021 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Product of UAE
V-38084CVS® Quality
Money Back Guarantee - Packaging
-
INGREDIENTS AND APPEARANCE
LONG ACTING COUGH SOFTGELS
dextromethorphan hydrobromide capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51316-778 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 15 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) ISOPROPYL ALCOHOL (UNII: ND2M416302) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYL GALLATE (UNII: 8D4SNN7V92) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SORBITAN (UNII: 6O92ICV9RU) Product Characteristics Color red Score no score Shape OVAL Size 14mm Flavor Imprint Code 778 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51316-778-23 1 in 1 CARTON 07/28/2021 1 20 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/28/2021 Labeler - CVS PHARMACY, INC (062312574)