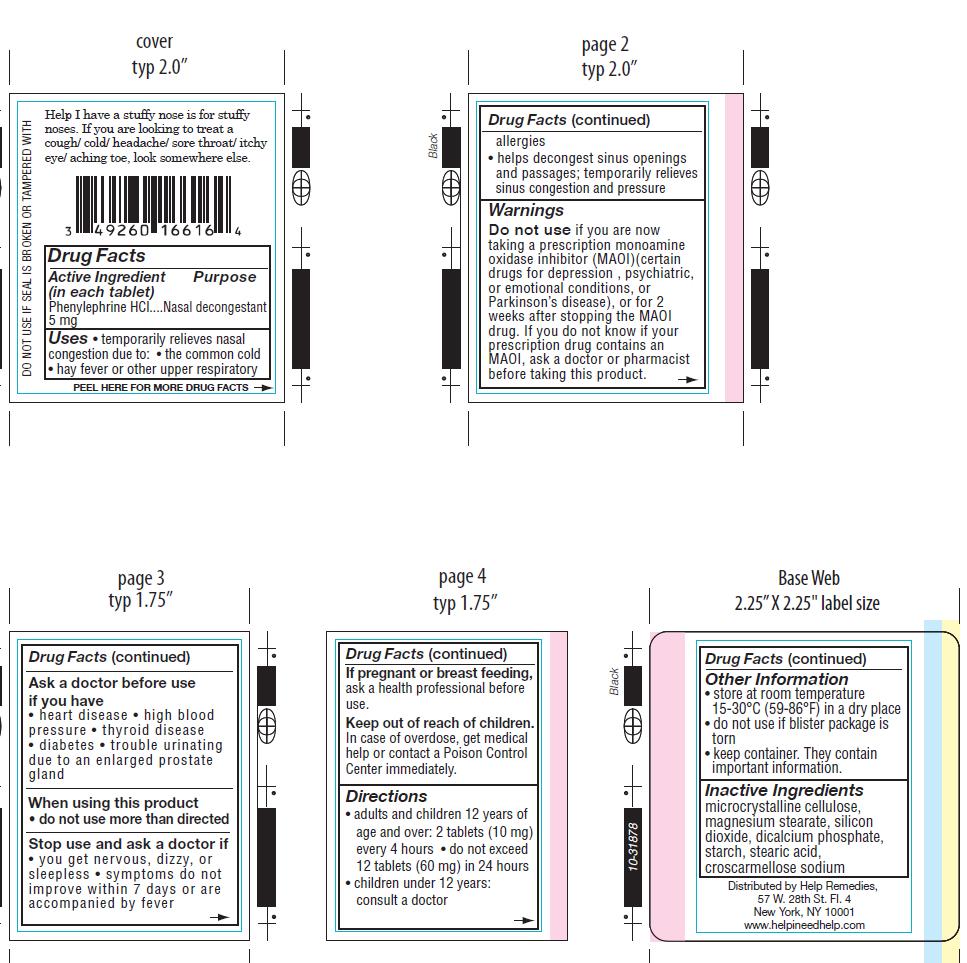
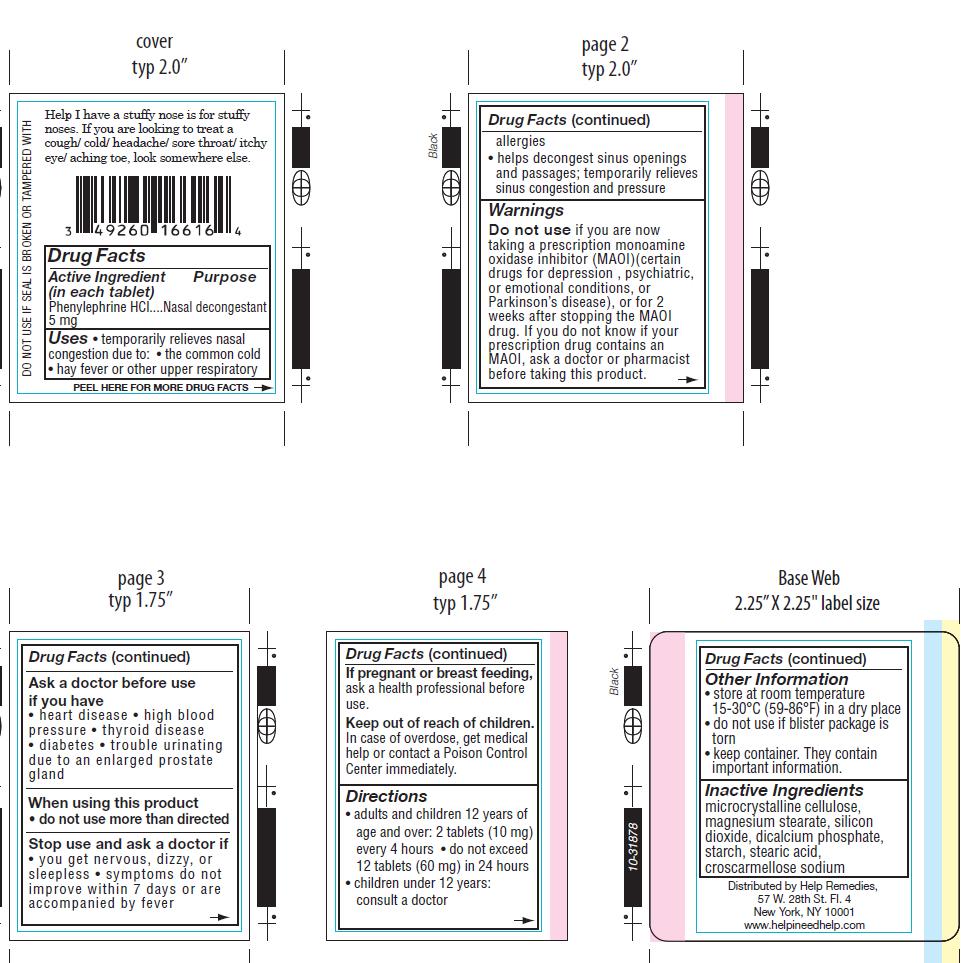
Label: HELP I HAVE A STUFFY NOSE- phenylephrine tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 49260-716-16, 49260-716-50 - Packager: Help Remedies, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 9, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI)(certain
drugs for depression , psychiatric, or emotional conditions, or Parkinson’s disease), or for 2
weeks after stopping the MAOI drug. If you do not know i your prescription drug contains an
MAOI, ask a doctor of pharmacist before taking this product.
Ask a doctor before use if you have
• heart disease • high blood pressure • thyroid disease • diabetes • trouble urinating due to
an enlarged prostate gland.
Stop use and ask a doctor if
• you get nervous, dizzy, or sleepless • symptoms do not improve within 7 days or are
accompanied by fever.
- DO NOT USE
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- DOSAGE & ADMINISTRATION
- INDICATIONS & USAGE
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HELP I HAVE A STUFFY NOSE
phenylephrine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49260-716 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white (white) Score no score Shape ROUND (round tablet) Size 7mm Flavor Imprint Code FR4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49260-716-50 50 in 1 BOTTLE 2 NDC:49260-716-16 16 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/01/2012 Labeler - Help Remedies, Inc. (050359682) Registrant - Help Remedies, Inc. (050359682)