Label: EPIPHANIQ- zincum iodatum solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 79813-204-11, 79813-204-21, 79813-204-51, 79813-204-52 - Packager: EpiphaniQ LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 29, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
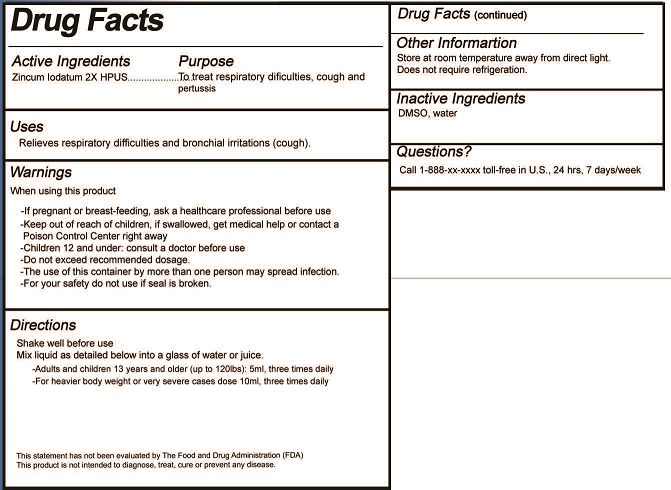
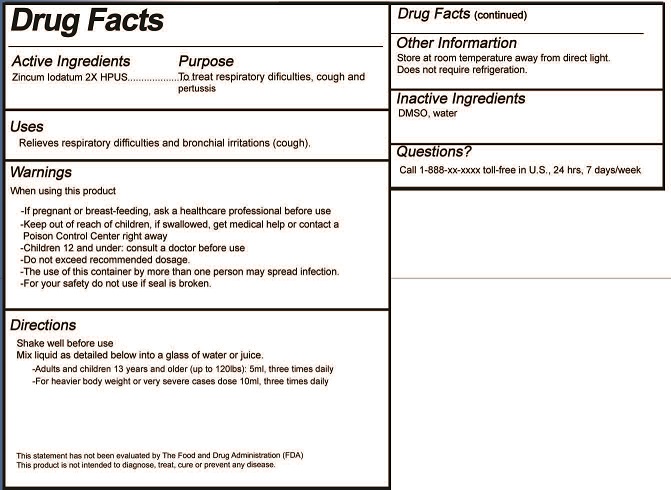
- ACTIVE INGREDIENT
- PURPOSE
- USES
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
Oral Liquid:
Shake well before use.
Mix liquid as detailed below into a glass of water or juice.
- Adults and children 13 years and older (up to 120lbs): 5ml, three times daily
- For heavier body weight or very severe cases dose 10ml, three times daily
Sachet:
Pour contents of sachets into a glass of water or juice as detailed below
- Adults and children 13 years and older (up to 120lbs): 1 sachet, three times daily
- For heavier body weight or very severe cases dose 2 sachets, three times daily
- INACTIVE INGREDIENTS
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EPIPHANIQ
zincum iodatum solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79813-204 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC IODIDE (UNII: 762R7A0O0B) (ZINC IODIDE - UNII:762R7A0O0B) ZINC IODIDE 2 [hp_X] in 100 mL Inactive Ingredients Ingredient Name Strength DIMETHYL SULFOXIDE (UNII: YOW8V9698H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79813-204-51 1 in 1 CARTON 07/23/2020 1 NDC:79813-204-11 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:79813-204-52 50 in 1 CARTON 07/23/2020 2 NDC:79813-204-21 5 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 07/23/2020 Labeler - EpiphaniQ LLC (117591249)