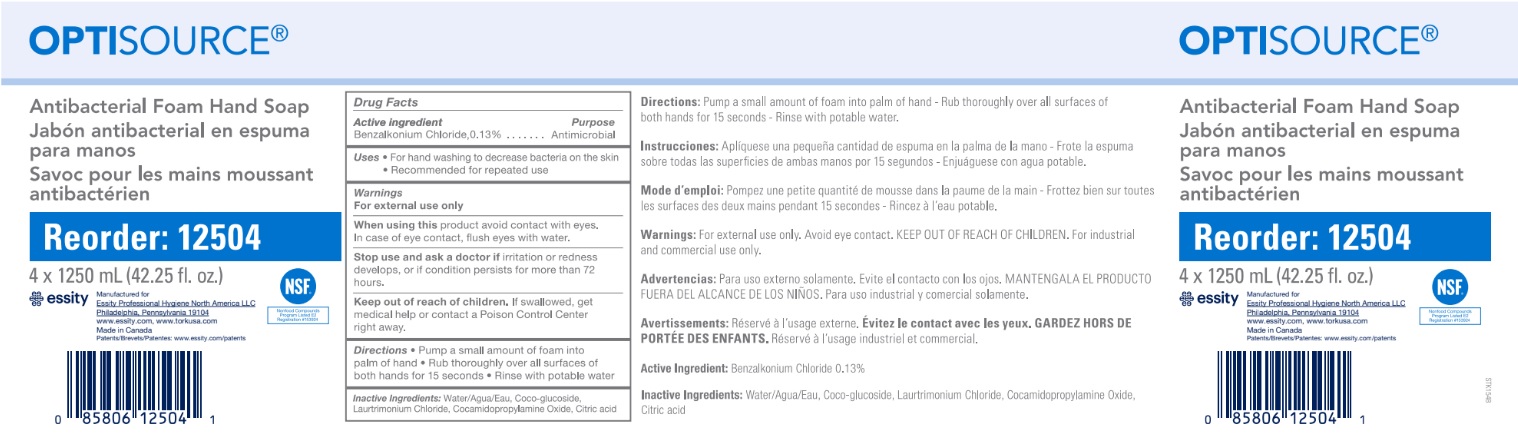
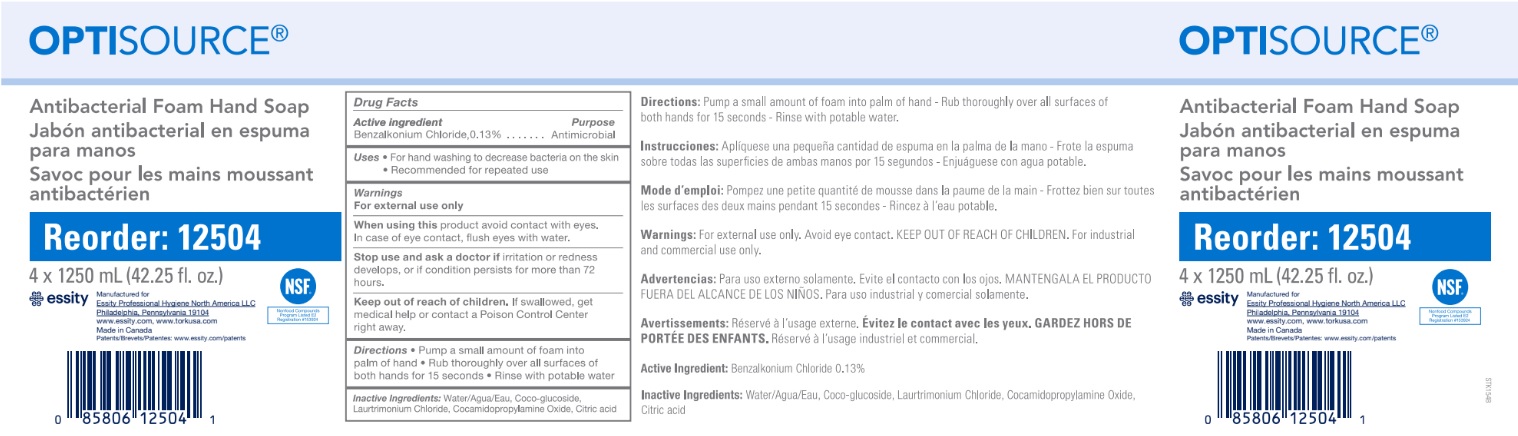
Label: OPTISOURCE ANTIBACTERIAL FOAM HAND- benzalkonium chloride soap
-
Contains inactivated NDC Code(s)
NDC Code(s): 64646-001-02 - Packager: Charlotte Products Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 4, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Warnings
- When using
- Stop using and ask doctor if
- Keep out of reach of children.
- Directions
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
OPTISOURCE ANTIBACTERIAL FOAM HAND
benzalkonium chloride soapProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64646-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) COCAMIDOPROPYLAMINE OXIDE (UNII: M4SL82J7HK) LAURTRIMONIUM CHLORIDE (UNII: A81MSI0FIC) COCO GLUCOSIDE (UNII: ICS790225B) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64646-001-02 4 in 1 CASE 12/22/2015 1 1250 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 11/03/2015 Labeler - Charlotte Products Ltd. (248293359) Registrant - Charlotte Products Ltd. (248293359) Establishment Name Address ID/FEI Business Operations Charlotte Products Ltd. 248293359 manufacture(64646-001)