Label: POLY-VI-SOL- vitamin a, vitamin d, .alpha.-tocopherol, thiamine, riboflavin, niacin, pyridoxine, and cyanocobalamin liquid
- NHRIC Code(s): 76413-337-01
- Packager: Central Texas Community Health Centers
- Category: DIETARY SUPPLEMENT
Drug Label Information
Updated October 20, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- HEALTH CLAIM
- Indication
-
• Rationale and Special Characteristics
- Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement has 9 important vitamins in a convenient daily dose. It is an excellent supplement for a child when transitioning to solid foods, during growth spurts and for the picky eater. Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement (without iron) is recommended for children who have sufficient iron in their diet or the possibility of too much.
The American Academy of Pediatrics (AAP) recommends 400 IU of supplemental vitamin D per day beginning in the first days of life for all breastfed and partially breastfed infants who do not receive at least 1 L of infant formula per day1.
Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement is does not contain ingredients derived from the most common food allergies: milk, eggs, peanuts, tree nuts, fish, shellfish, soy or wheat.
Refrigeration is not required.
- Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement has 9 important vitamins in a convenient daily dose. It is an excellent supplement for a child when transitioning to solid foods, during growth spurts and for the picky eater. Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement (without iron) is recommended for children who have sufficient iron in their diet or the possibility of too much.
-
• Nutrients
Per 1 mL % DV Infants % DV Children under 4 years o Note: Enfamil Poly-Vi-Sol Liquid Multivitamin Supplement does not include folic acid Vitamin A, IU 750 50 30 Vitamin D, IU 400 100 100 Vitamin E, IU 5 100 50 Vitamin C, mg 35 100 88 Thiamin, mg 0.5 100 71 Riboflavin, mg 0.6 100 75 Niacin, mg 8 100 89 Vitamin B6, mg 0.4 100 57 Vitamin B12, mcg 2 100 67 - • Osmolality
- • Product Form
-
• Composition
- Ingredients: Glycerin, water, ascorbic acid, vitamin E succinate, niacinamide, polysorbate 80, ferrous sulfate (stabilizer for vitamin B12), caramel color, vitamin A palmitate, thiamin hydrochloride, riboflavin-5-phosphate sodium, vitamin D3, vitamin B6 hydrochloride, natural and artificial flavor, sulfuric acid (antioxidant for iron), vitamin B12, sulfites.
-
• Administration
- Drops: Now includes an easy-to-use syringe. The syringe is marked with 0.25 mL, 0.5 mL, 0.75 mL and 1.0 mL dose lines. To administer directly, place the syringe in the child's mouth with the tip against the inside of the cheek. A firm pressure on the syringe bulb will deliver the proper dose. A slight excess will remain in the syringe. If preferred, the drops may be slowly mixed with formula, juice, cereal or other food and fed within 1 hour.
- • Storage
- • Precautions
-
• References
- ○
- 1. Wagner CL, Greer FR, and the American Academy of Pediatrics Section on Breastfeeding and Committee on Nutrition. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics. 2008;122:1142-1152.
- 2.
- Testing was conducted in September 2009 by Mead Johnson Nutrition, Quality Control.
- HEALTH CLAIM
-
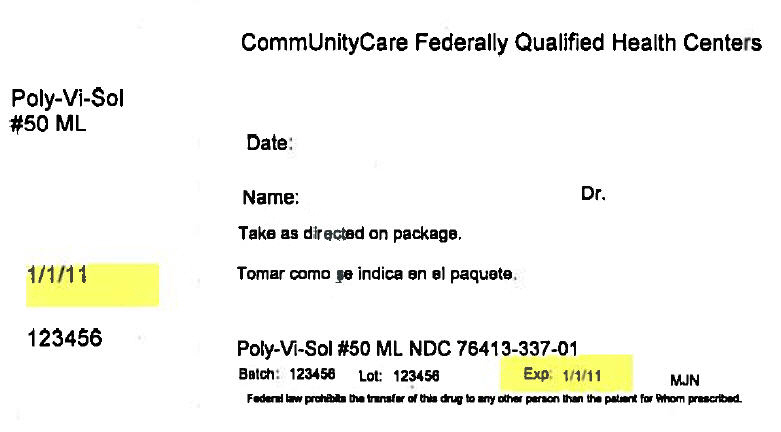
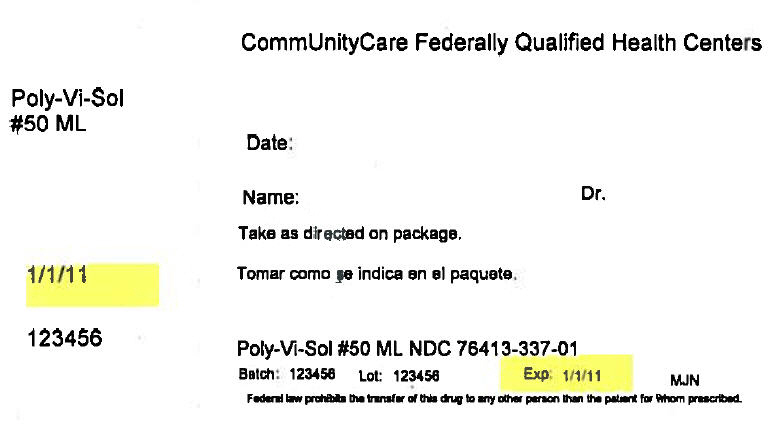
PRINCIPAL DISPLAY PANEL - 50 ML Bottle Box Label
CommUnityCare Federally Qualified Health Centers
Poly-Vi-Sol
#50 MLDate:
Name:
Dr.Take as directed on package.
1/1/11
123456
Poly-Vi-Sol #50 ML NDC 76413-337-01
Batch: 123456
Lot: 123456
Exp: 1/1/11
MJNFederal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
-
INGREDIENTS AND APPEARANCE
POLY-VI-SOL
vitamin a, vitamin d, .alpha.-tocopherol, thiamine, riboflavin, niacin, pyridoxine, and cyanocobalamin liquidProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:76413-337 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A (UNII: 81G40H8B0T) (Vitamin A - UNII:81G40H8B0T) Vitamin A 750 [iU] in 1 mL Vitamin D (UNII: 9VU1KI44GP) (Cholecalciferol - UNII:1C6V77QF41, Vitamin D - UNII:9VU1KI44GP) Vitamin D 400 [iU] in 1 mL .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) (.ALPHA.-TOCOPHEROL - UNII:H4N855PNZ1) .ALPHA.-TOCOPHEROL 5 [iU] in 1 mL THIAMINE (UNII: X66NSO3N35) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 0.5 mg in 1 mL Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 0.6 mg in 1 mL Niacin (UNII: 2679MF687A) (Niacin - UNII:2679MF687A) Niacin 8 mg in 1 mL PYRIDOXINE (UNII: KV2JZ1BI6Z) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 0.4 mg in 1 mL CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 2 ug in 1 mL Inactive Ingredients Ingredient Name Strength glycerin (UNII: PDC6A3C0OX) water (UNII: 059QF0KO0R) polysorbate 80 (UNII: 6OZP39ZG8H) sulfuric acid (UNII: O40UQP6WCF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:76413-337-01 1 in 1 BOX 1 50 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 01/01/2009 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 REPACK, RELABEL Establishment Name Address ID/FEI Business Operations Mead Johnson & Company, LLC 006370092 MANUFACTURE