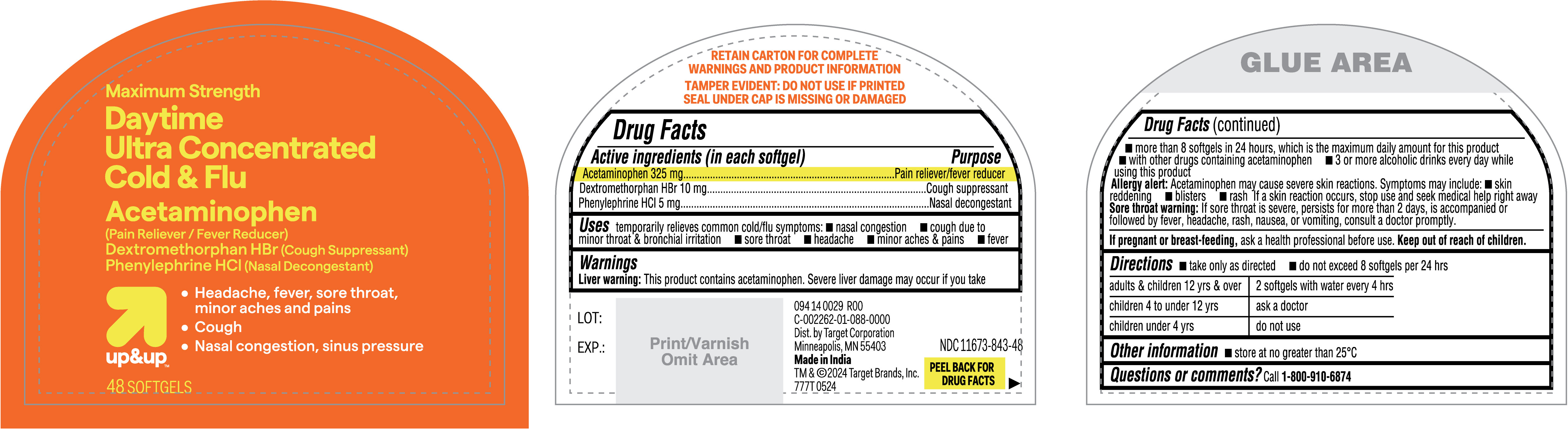
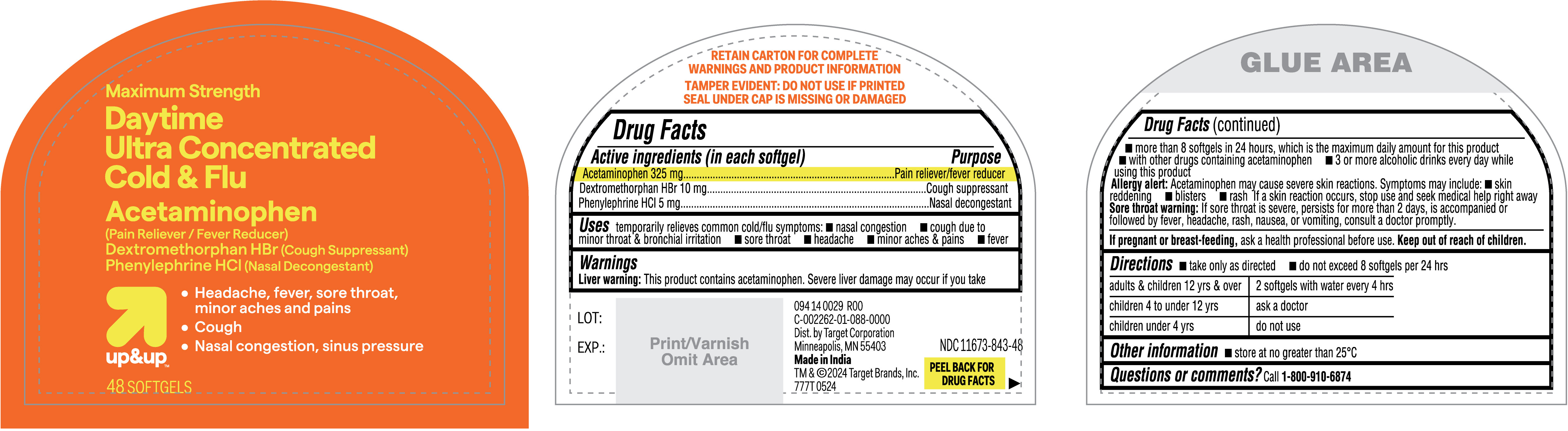
Label: DAYTIME ULTRA CONCENTRATED COLD AND FLU- acetaminophen, dextromethrophan hbr, phenylephrine hcl capsule, liquid filled
- NDC Code(s): 11673-843-48
- Packager: TARGET CORPORATION
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 26, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each softgel)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 8 softgels in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to enlarged prostate gland
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Stop use and ask a doctor if
- you get nervous, dizzy or sleepless
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DAYTIME ULTRA CONCENTRATED COLD AND FLU
acetaminophen, dextromethrophan hbr, phenylephrine hcl capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11673-843 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg Inactive Ingredients Ingredient Name Strength SHELLAC (UNII: 46N107B71O) GLYCERIN (UNII: PDC6A3C0OX) SORBITAN (UNII: 6O92ICV9RU) GELATIN (UNII: 2G86QN327L) MICA (UNII: V8A1AW0880) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) POVIDONE (UNII: FZ989GH94E) SORBITOL SOLUTION (UNII: 8KW3E207O2) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C YELLOW NO. 6 ALUMINUM LAKE (UNII: GYP6Z2JR6Q) Product Characteristics Color orange Score no score Shape OVAL Size 15mm Flavor Imprint Code 151 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11673-843-48 1 in 1 CARTON 06/30/2024 1 48 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 06/30/2024 Labeler - TARGET CORPORATION (006961700) Registrant - TIME CAP LABORATORIES INC (037005209) Establishment Name Address ID/FEI Business Operations MARKSANS PHARMA LTD 925822975 manufacture(11673-843)