Label: ARIELLA NAIL REPAIR PEN- tolnaftate liquid
- NDC Code(s): 83364-006-01
- Packager: YITONGBADA (SHENZHEN) INTERNATIONAL TRADE CO., LTD
- This is a repackaged label.
- Source NDC Code(s): 84010-003
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 17, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
PRINCIPAL DISPLAY PANEL
 Drug Facts
Drug Facts
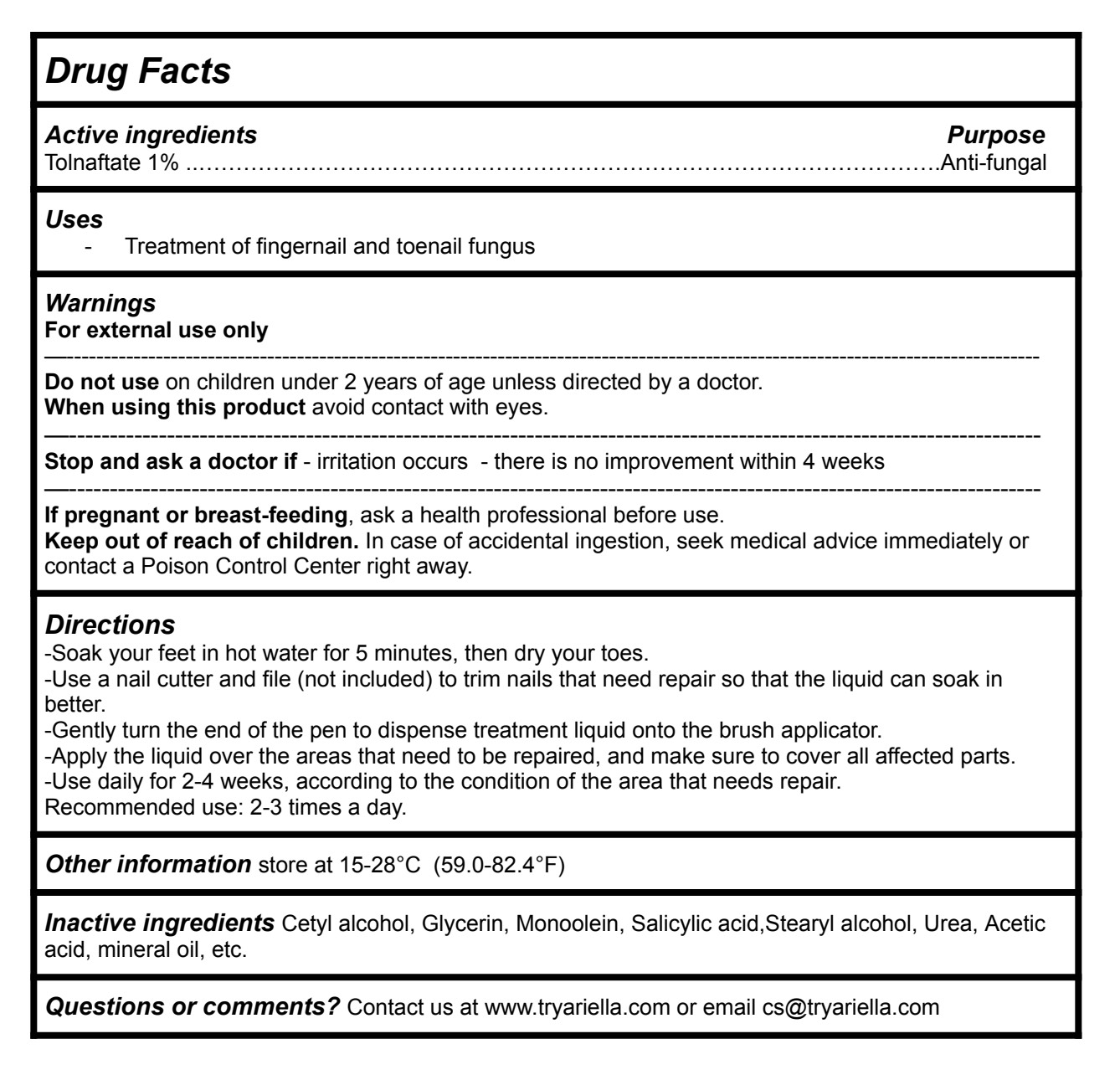
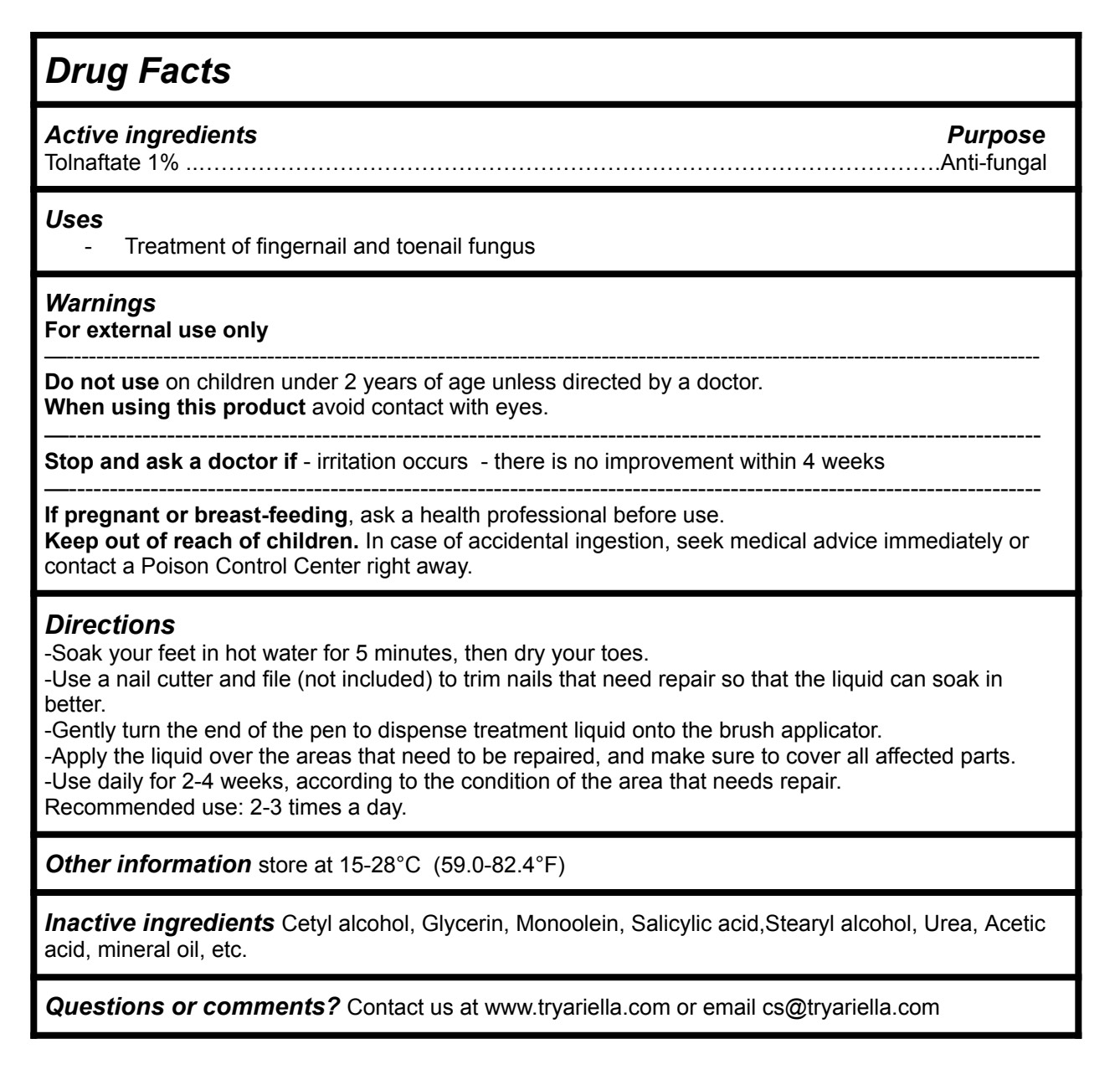
Active ingredients Purpose
Tolnaftate 1%............................................................................................Anti-fungal
Uses
-Treatment of fingernail and toenail fungus
Directions
-Soak your feet in hot water for 5 minutes, then dry your toes.
-Use a nail cutter and file (not included) to trim nails that need repair so that the liquid can soak in better.
-Gently turn the end of the pen to dispense treatment liquid onto the brush applicator.
-Apply the liquid over the areas that need to be repaired, and make sure to cover all affected parts.
-Use daily for 2-4 weeks, according to the condition of the area that needs repair. Recommended use: 2-3 times a day.
-
INGREDIENTS AND APPEARANCE
ARIELLA NAIL REPAIR PEN
tolnaftate liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83364-006(NDC:84010-003) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) SALICYLIC ACID (UNII: O414PZ4LPZ) GLYCERYL MONOOLEATE (UNII: C4YAD5F5G6) ACETIC ACID (UNII: Q40Q9N063P) CETYL ALCOHOL (UNII: 936JST6JCN) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) GLYCERIN (UNII: PDC6A3C0OX) UREA (UNII: 8W8T17847W) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83364-006-01 1 in 1 BOX 06/24/2024 1 4 in 1 TUBE 1 3 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 06/24/2024 Labeler - YITONGBADA (SHENZHEN) INTERNATIONAL TRADE CO., LTD (725220463)