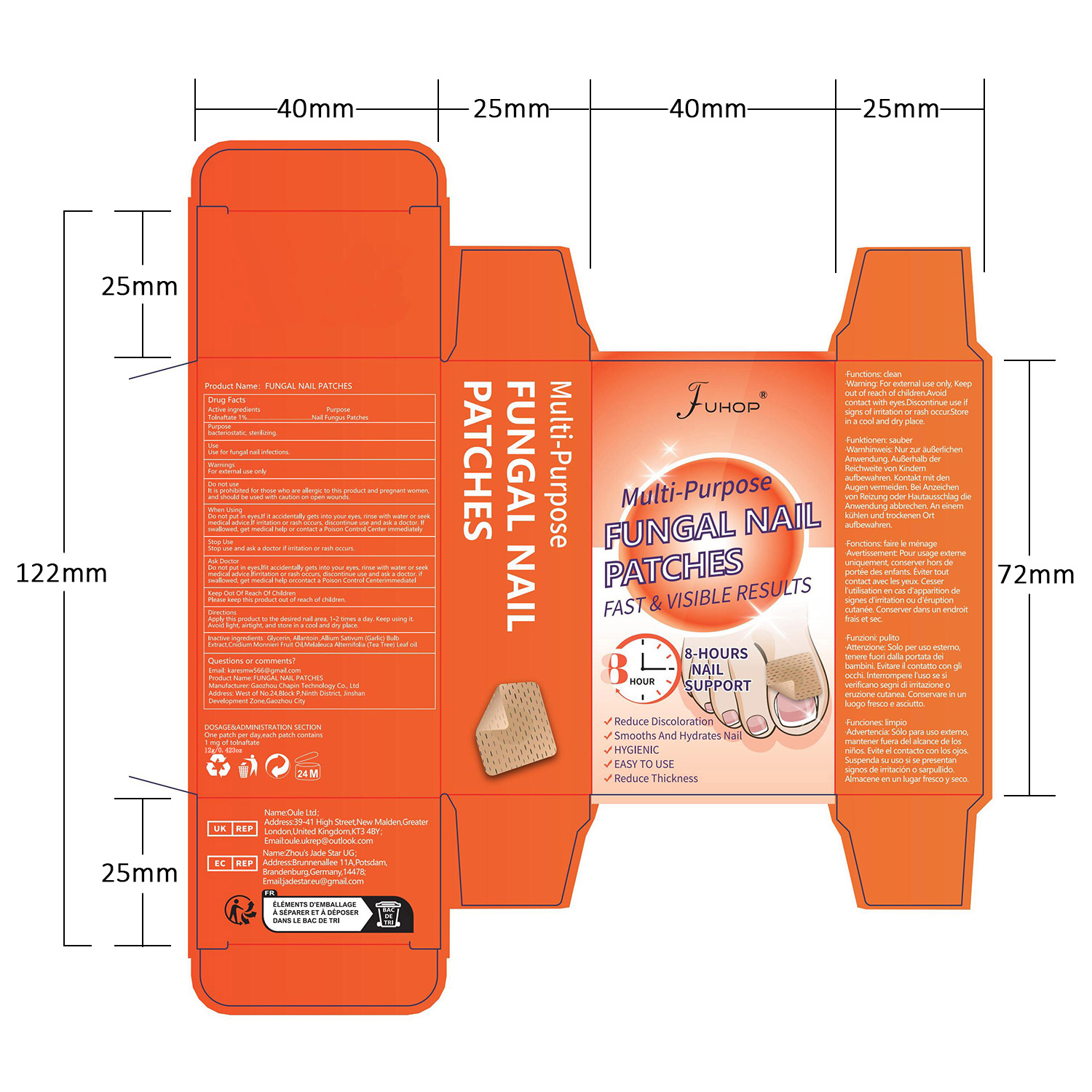
Label: FUHOP FUNGAL NAIL PATCHES- fungal nail patches patch
- NDC Code(s): 84168-035-01
- Packager: Gaozhou Chapin Technology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 10, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Ask Doctor
- Keep Out Of Reach Of Children
- Inactive ingredients
- DOSAGE & ADMINISTRATION SECTION
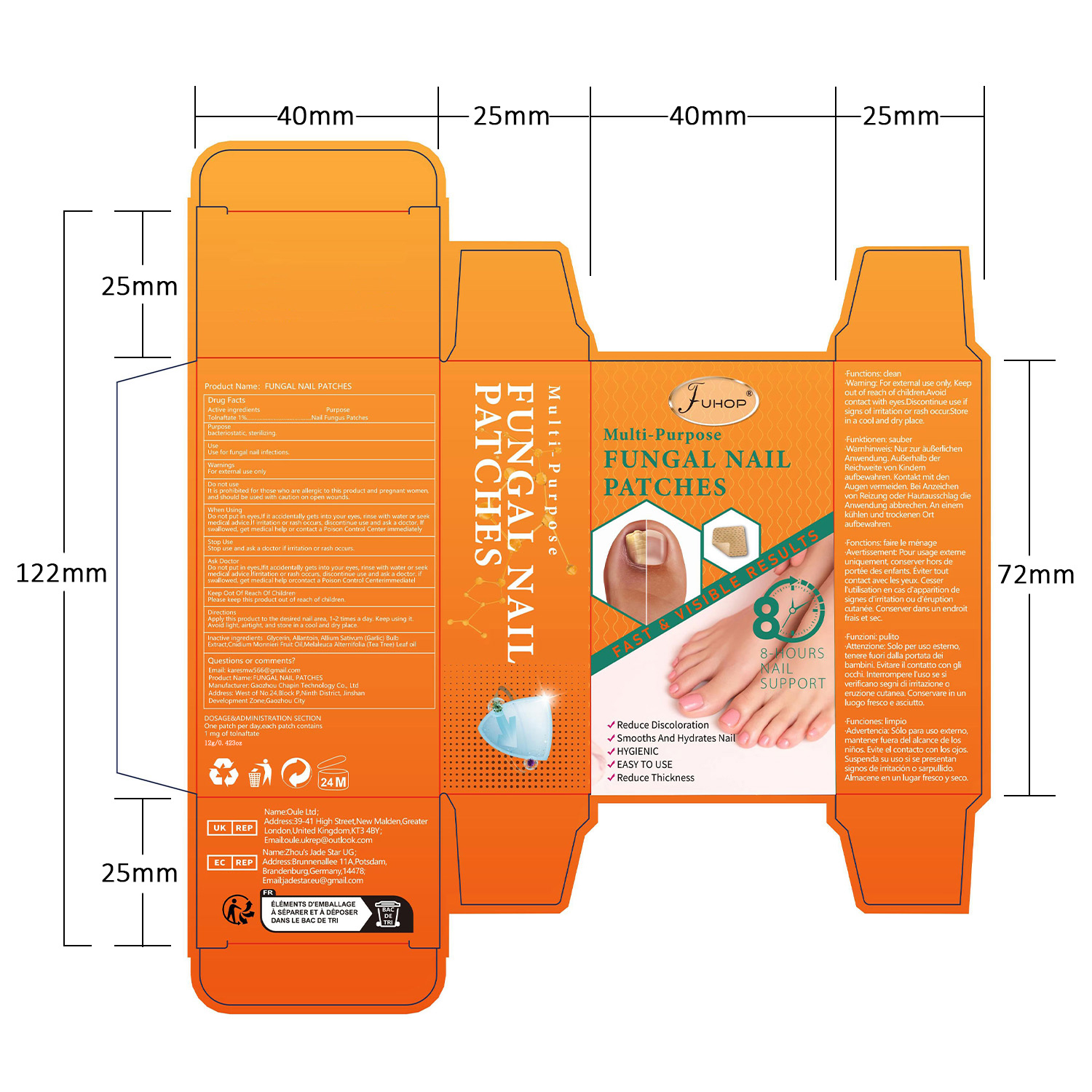
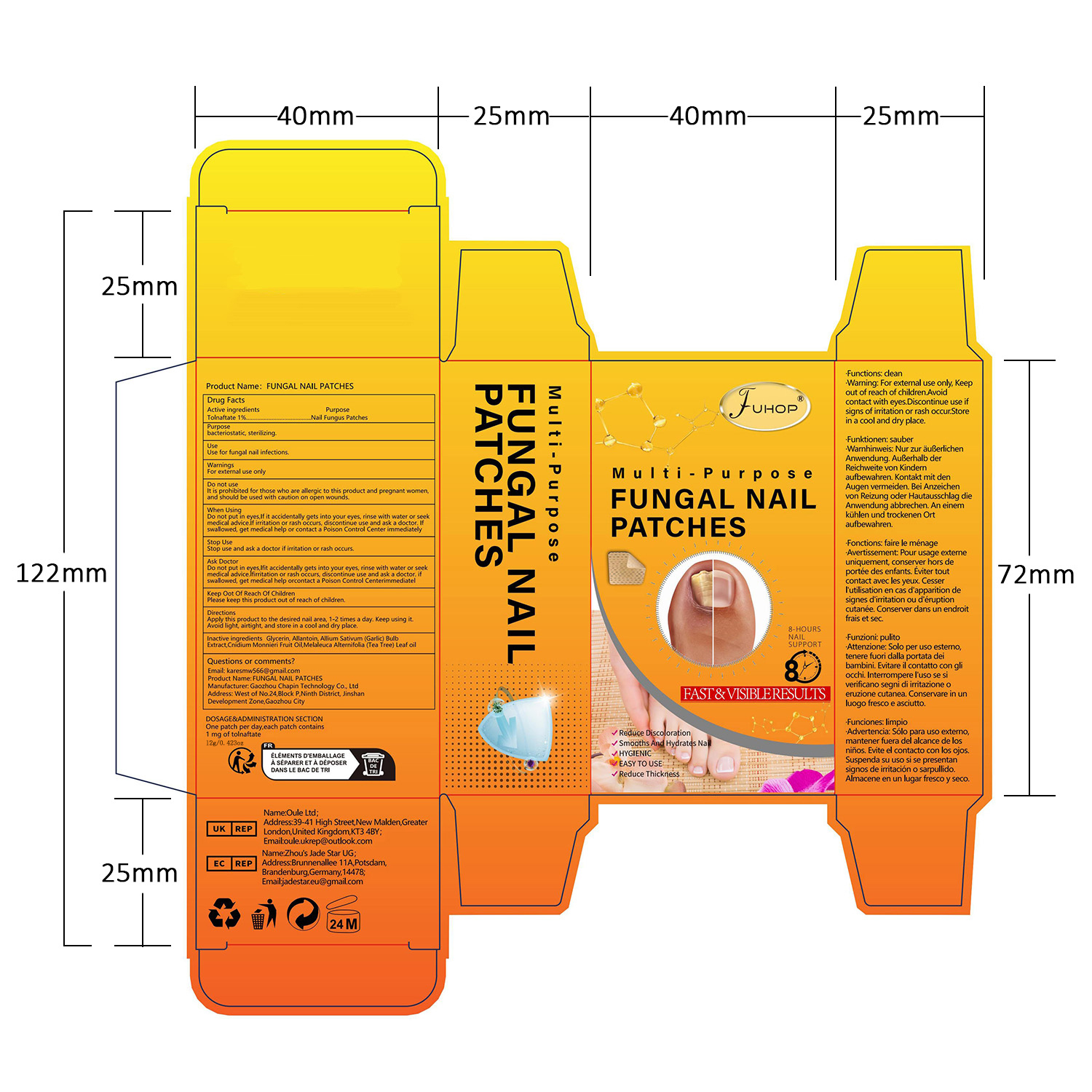
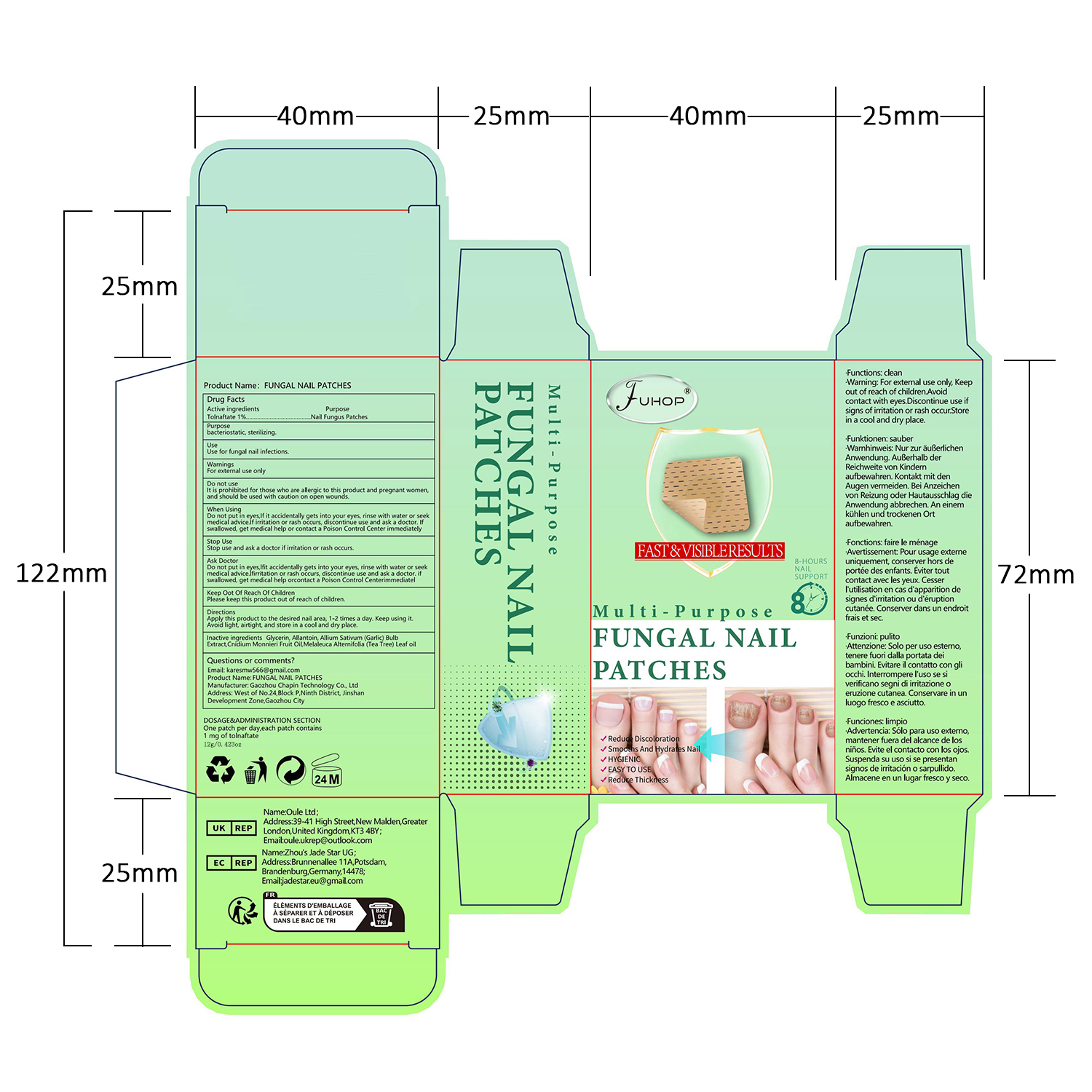
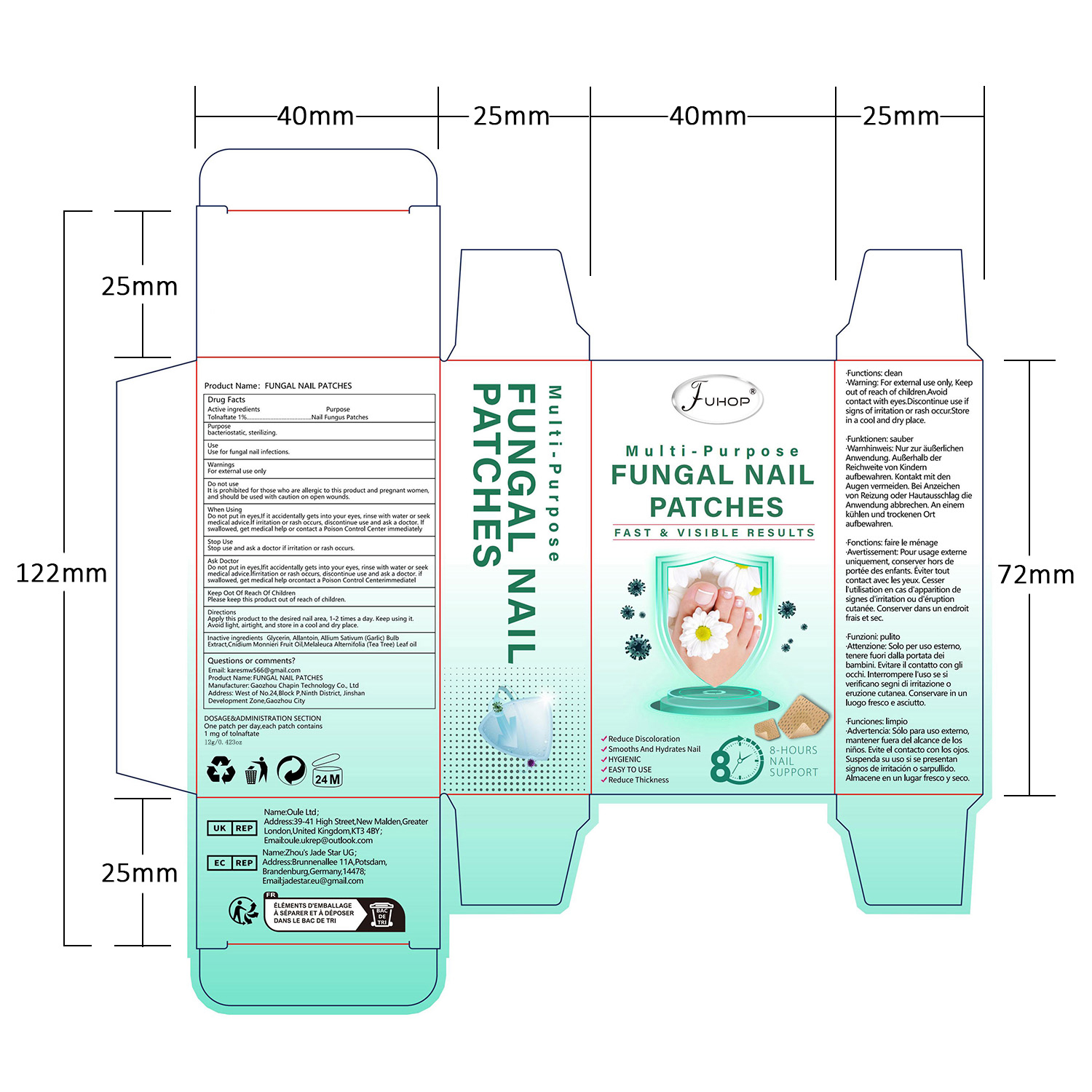
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FUHOP FUNGAL NAIL PATCHES
fungal nail patches patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84168-035 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 1 g in 100 g Inactive Ingredients Ingredient Name Strength GARLIC (UNII: V1V998DC17) MELALEUCA ALTERNIFOLIA (TEA TREE) LEAF OIL (UNII: VIF565UC2G) ALLANTOIN (UNII: 344S277G0Z) CNIDIUM MONNIERI FRUIT OIL (UNII: JK0MS9P8YL) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84168-035-01 12 g in 1 PATCH; Type 0: Not a Combination Product 06/10/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 06/10/2024 Labeler - Gaozhou Chapin Technology Co., Ltd (715573719) Establishment Name Address ID/FEI Business Operations Gaozhou Chapin Technology Co., Ltd 715573719 manufacture(84168-035)