Label: KROGER EXTRA STRENGTH- aluminum hydroxide and magnesium carbonate tablet, chewable
- NDC Code(s): 30142-610-00
- Packager: THE KROGER CO.,
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 29, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
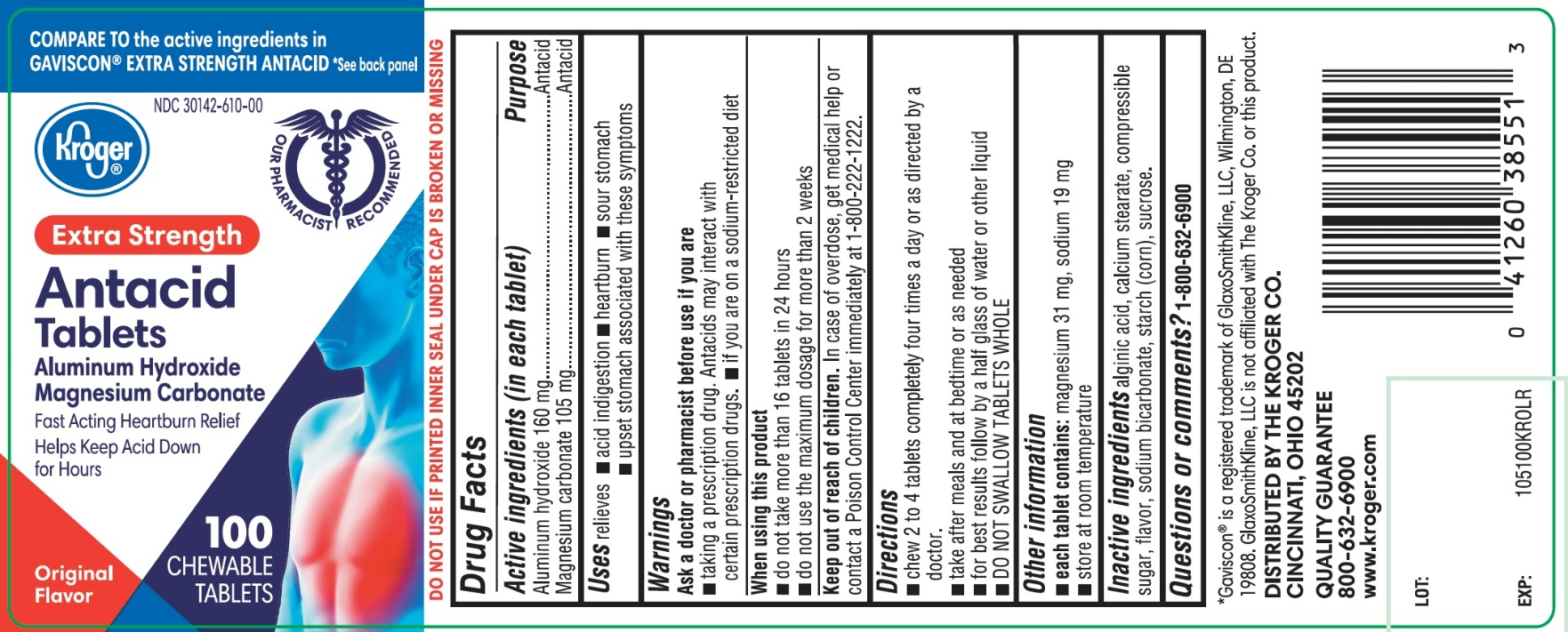
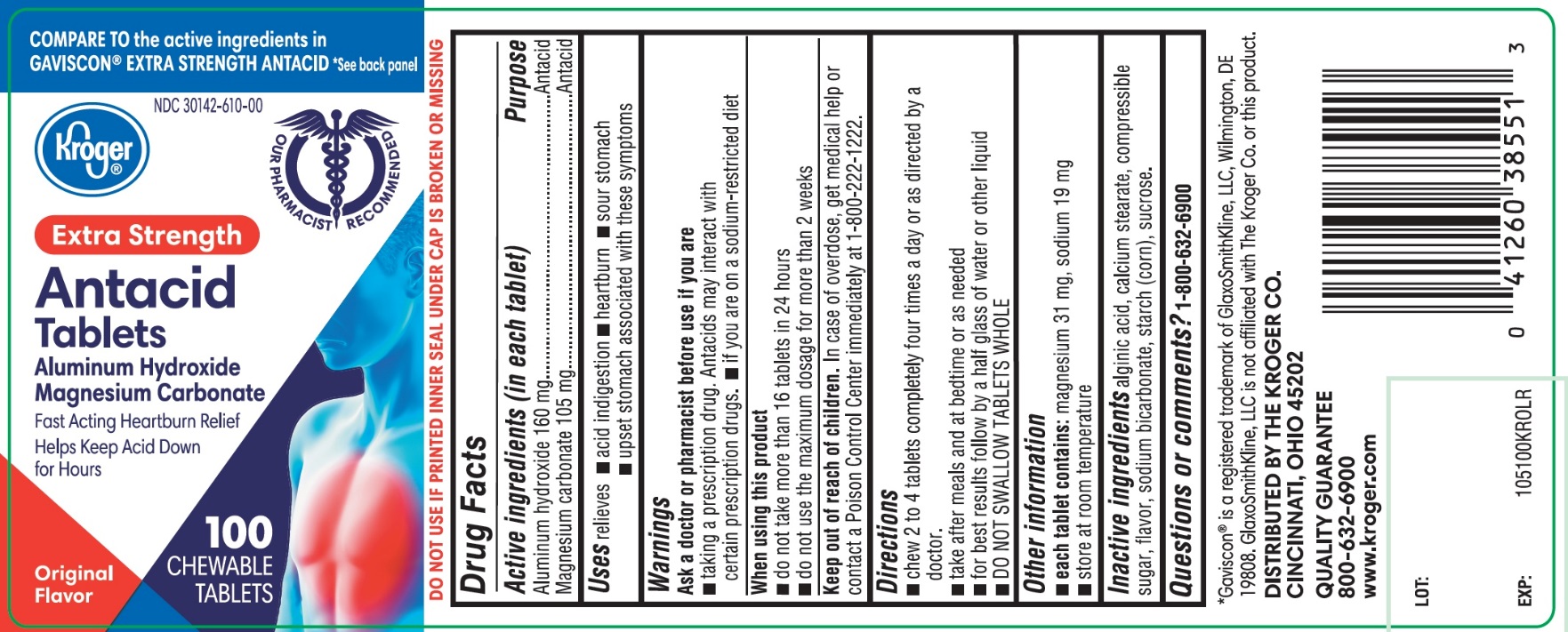
- Active ingredients (in each tablet)
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
-
Principal Display Panel
Kroger ®
NDC 30142-610-00
COMPARE TO the active ingredients GAVISCON®EXTRA STRENGTH ANTACID
*See back panel
Antacid Tablets
EXTRA STRENGTH
Aluminum Hydroxide, 160 mg
Magnesium Carbonate, 105 mg
Fast Acting Heartburn Relief, Helps Keep Acid Down for Hours
- OUR PHARMACISTS RECOMMEND
100 CHEWABLE TABLETS
Original Flavor
DISTRIBUTED BY THE KROGER CO.
CINCINNATI, OHIO 45202
QUALITY GUARANTEE
800-632-6900 www.kroger.com
GLUTEN FREE
DO NOT USE IF PRINTED INNER SEAL UNDER CAP IS BROKEN OR MISSING
*GAVISCON ®IS A REGISTERED TRADEMARK OF GLAXOSMITHKLINE LLC, WILMINGTON, DE 19808. GLAXOSMITHKLINE LLC IS NOT AFFLIATED WITH THE KROGER CO. OR THIS PRODUCT.
-
INGREDIENTS AND APPEARANCE
KROGER EXTRA STRENGTH
aluminum hydroxide and magnesium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:30142-610 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) (ALUMINUM HYDROXIDE - UNII:5QB0T2IUN0) ALUMINUM HYDROXIDE 160 mg MAGNESIUM CARBONATE (UNII: 0E53J927NA) (CARBONATE ION - UNII:7UJQ5OPE7D) MAGNESIUM CARBONATE 105 mg Inactive Ingredients Ingredient Name Strength ALGINIC ACID (UNII: 8C3Z4148WZ) CALCIUM STEARATE (UNII: 776XM7047L) CORN SYRUP (UNII: 9G5L16BK6N) SODIUM BICARBONATE (UNII: 8MDF5V39QO) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) Product Characteristics Color white Score no score Shape ROUND Size 17mm Flavor BUTTERSCOTCH (Original) Imprint Code RP105 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:30142-610-00 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/16/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 05/16/2019 Labeler - THE KROGER CO., (006999528)