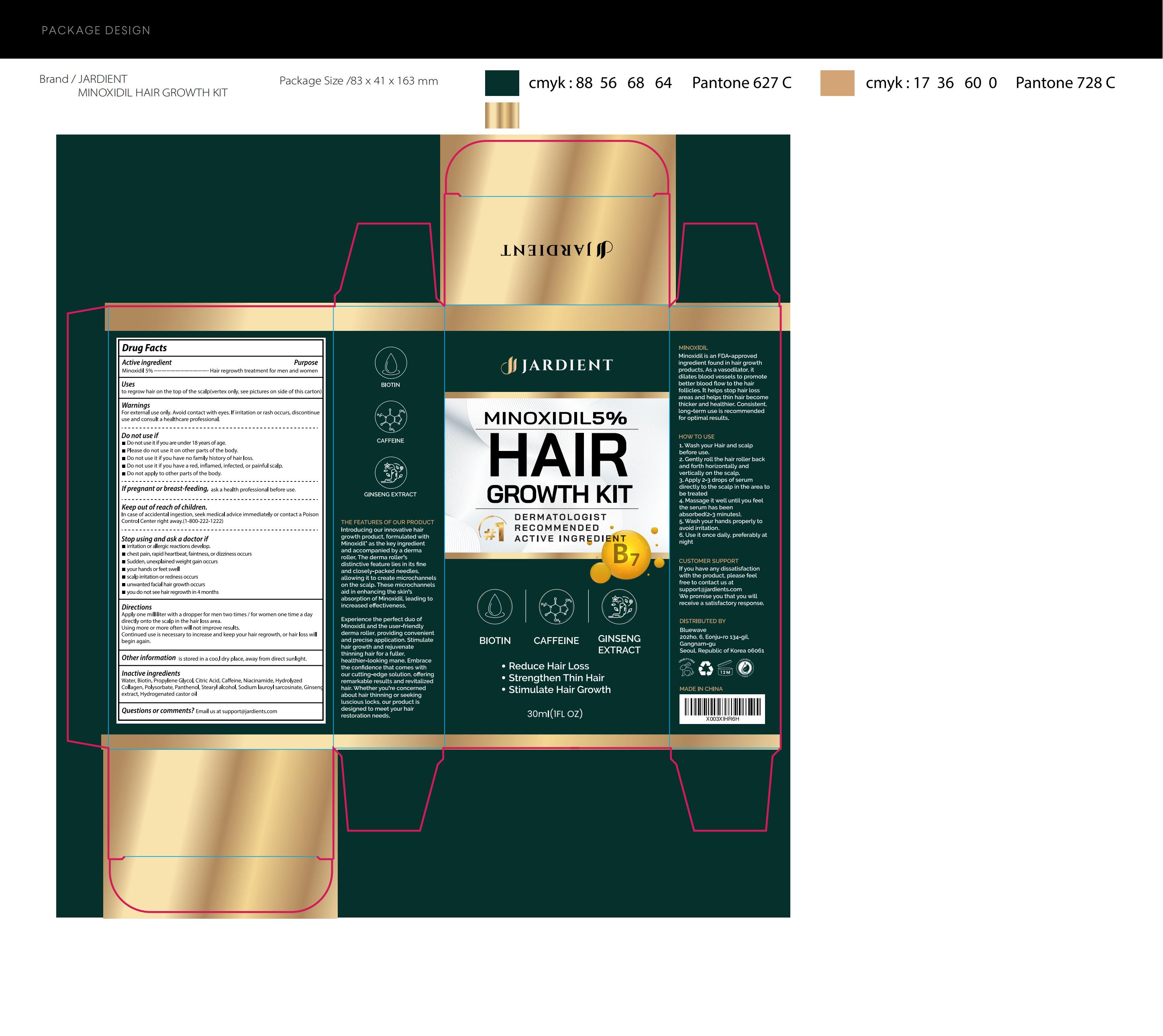
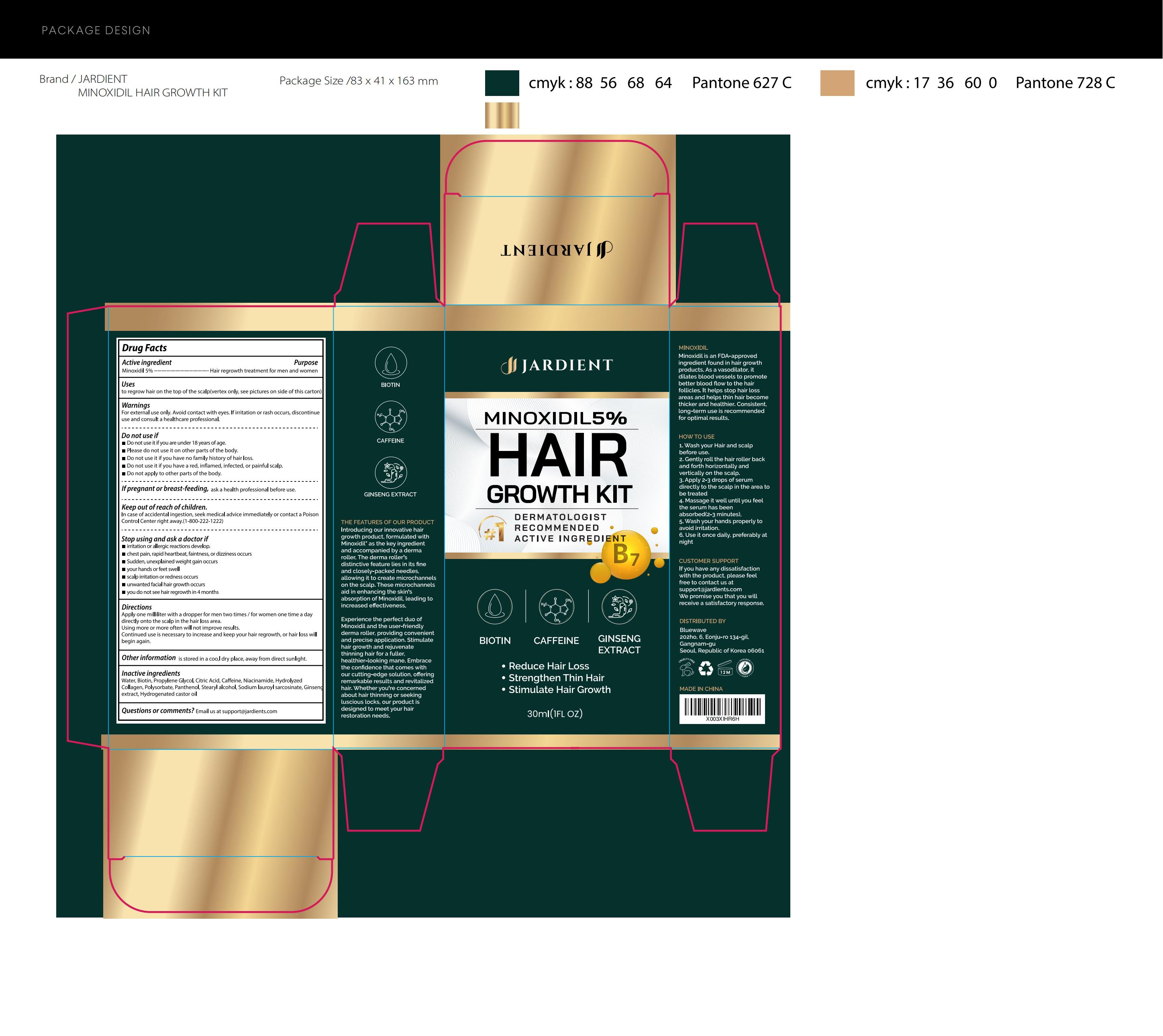
Label: JARDIENT 5% MINOXIDIL HAIR GROWTH SERUM- 5% minoxidil hair growth serum liquid
- NDC Code(s): 83596-003-01
- Packager: Aramode
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 10, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
-
Stop Use
Stop using and ask a doctor if
irritation or allergic reactions develop.
chest pain, rapid heartbeat, faintness, or dizziness occurs
Sudden, unexplained weight gain occurs
your hands or feet swell
scalp irritation or redness occurs
unwanted facial hair growth occurs
you do not see hair regrowth in 4 months -
Ask Doctor
Stop using and ask a doctor if
irritation or allergic reactions develop.
chest pain, rapid heartbeat, faintness, or dizziness occurs
Sudden, unexplained weight gain occurs
your hands or feet swell
scalp irritation or redness occurs
unwanted facial hair growth occurs
you do not see hair regrowth in 4 months - Keep Oot Of Reach Of Children
- Directions
- Other information
- Inactive ingredients
- Questions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
JARDIENT 5% MINOXIDIL HAIR GROWTH SERUM
5% minoxidil hair growth serum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83596-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 5 g in 100 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) NIACINAMIDE (UNII: 25X51I8RD4) POLYSORBATE 21 (UNII: 59IO08SBZU) SODIUM LAUROYL SARCOSINATE (UNII: 632GS99618) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) WATER (UNII: 059QF0KO0R) BIOTIN (UNII: 6SO6U10H04) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CAFFEINE (UNII: 3G6A5W338E) COLLAGEN ALPHA-1(I) CHAIN BOVINE (UNII: FB3DQM32F2) PANTHENOL (UNII: WV9CM0O67Z) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) ASIAN GINSENG (UNII: CUQ3A77YXI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83596-003-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/19/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 11/19/2023 Labeler - Aramode (963192477) Establishment Name Address ID/FEI Business Operations Aramode 963192477 manufacture(83596-003)