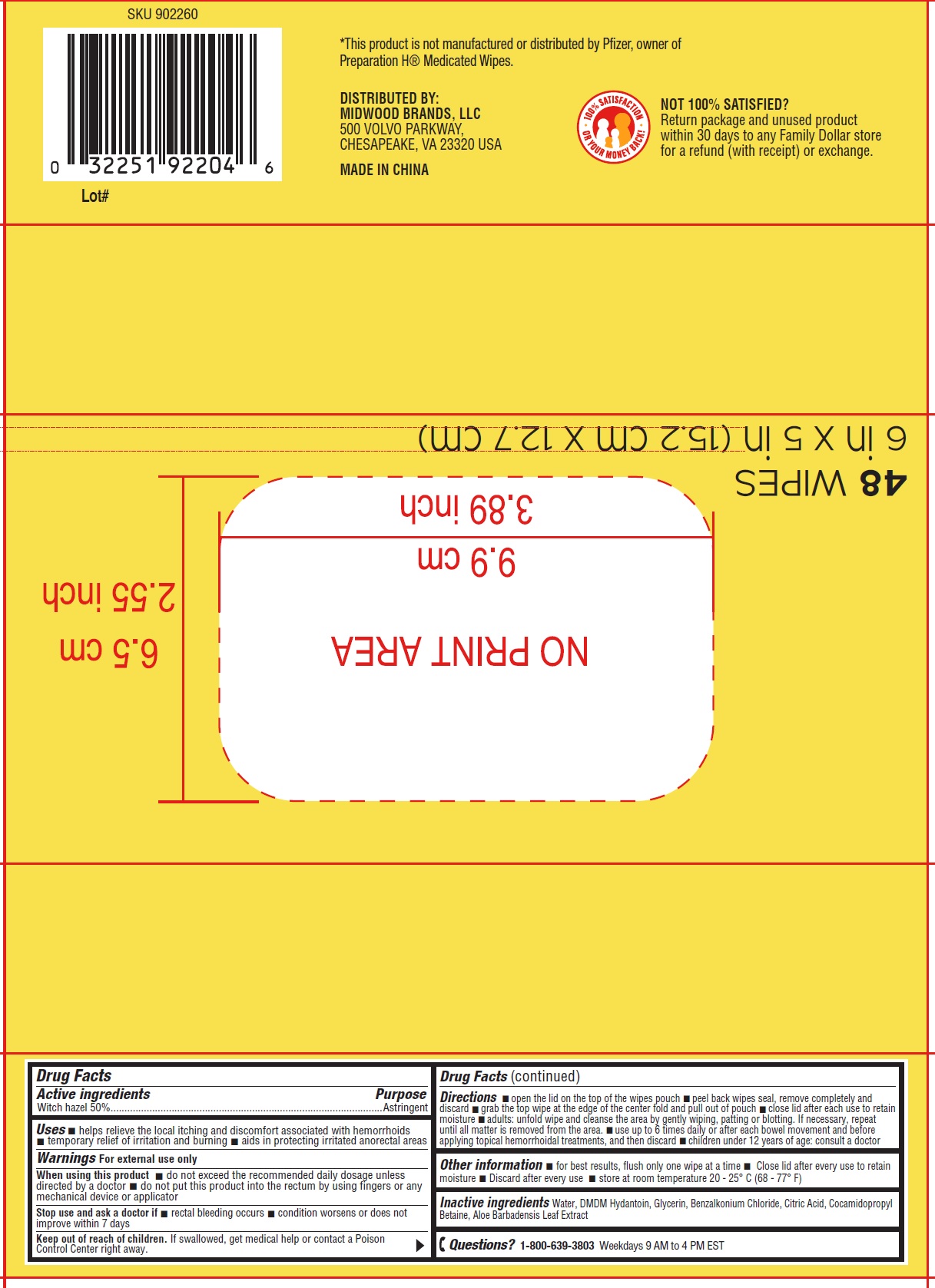
Label: MEDICATED WIPES HEMORROIDAL WIPES WITH WITCH HAZEL- witch hazel cloth
- NDC Code(s): 72932-009-00
- Packager: Wuhan Zonsen Medical Products Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated February 26, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
- Uses
-
Warnings
For external use only
When using this product
- do not exceed the recommended daily dosage unless directed by a doctor
- do not put this product into the rectum by using fingers or any mechanical device or applicator
-
Directions
- open the lid on the top of the wipes pouch
- peel back wipes seal, remove completely and discard
- grab the top wipe at the edge of the center fold and pull out of pouch
- close lid after each use to retain moisture
- adults: unfold wipe and cleanse the area by gently wiping, patting or blotting. If necessary, repeat until all matter is removed from the area.
- se up to 6 times daily or after each bowel movement and before applying topical hemorrhoidal treatments, and then discard
- children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Questions?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
MEDICATED WIPES HEMORROIDAL WIPES WITH WITCH HAZEL
witch hazel clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72932-009 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WITCH HAZEL (UNII: 101I4J0U34) (WITCH HAZEL - UNII:101I4J0U34) WITCH HAZEL 500 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DMDM HYDANTOIN (UNII: BYR0546TOW) GLYCERIN (UNII: PDC6A3C0OX) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72932-009-00 48 in 1 BAG 01/01/2024 1 1.4717 mL in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M015 01/01/2024 Labeler - Wuhan Zonsen Medical Products Co., Ltd. (554474517) Registrant - Wuhan Zonsen Medical Products Co., Ltd. (554474517) Establishment Name Address ID/FEI Business Operations Wuhan Zonsen Medical Products Co., Ltd. 554474517 manufacture(72932-009)