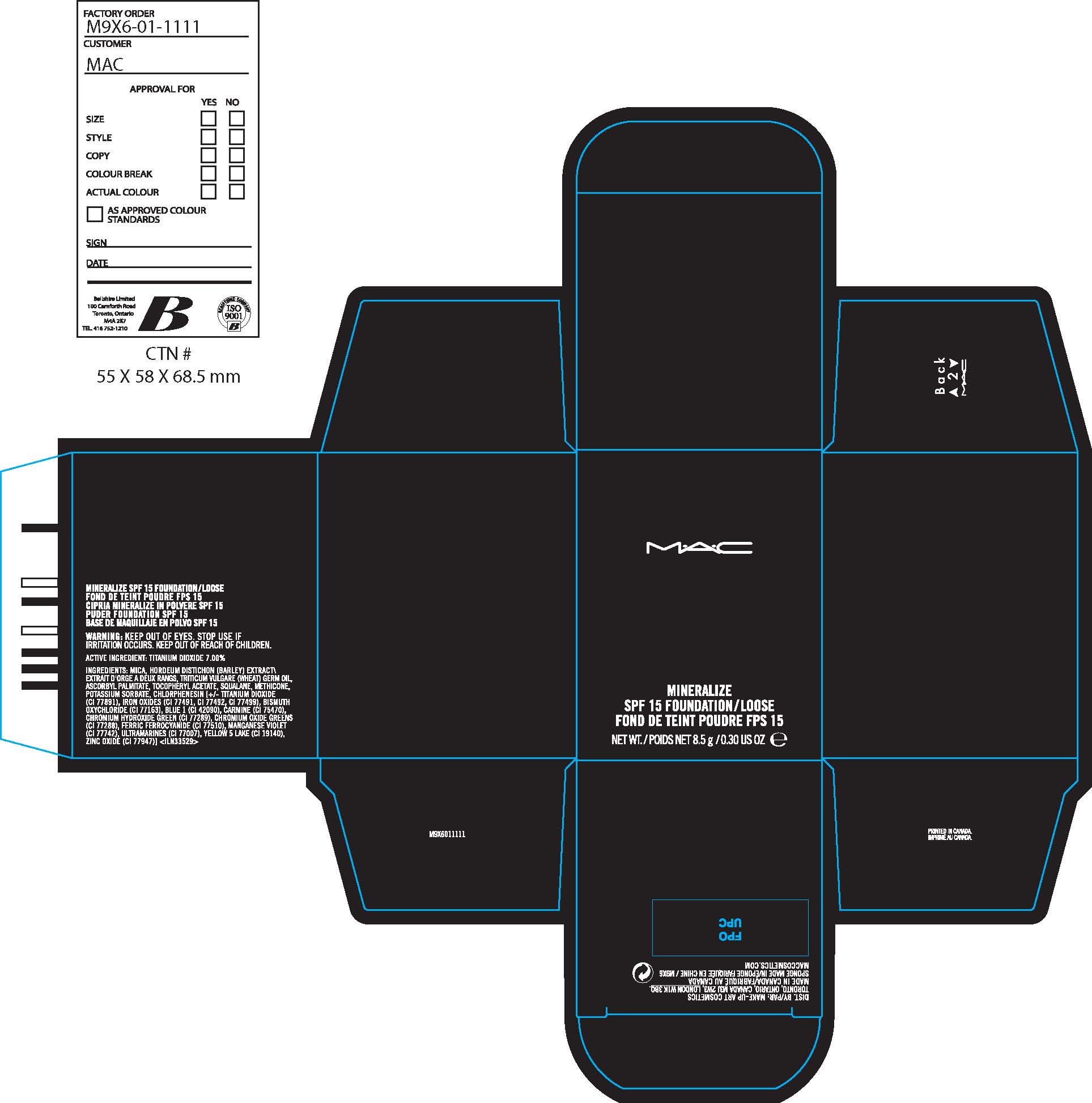
Label: MINERALIZE SPF 15 FOUNDATION LOOSE- titanium dioxide powder
-
Contains inactivated NDC Code(s)
NDC Code(s): 40046-0031-1, 40046-0031-2 - Packager: MAKEUP ART COSMETICS
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 12, 2011
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
INACTIVE INGREDIENTS: MICA [] HORDEUM DISTICHON (BARLEY) EXTRACT\EXTRAIT D'ORGE A DEUX RANGS [] TRITICUM VULGARE (WHEAT) GERM OIL [] ASCORBYL PALMITATE [] TOCOPHERYL ACETATE [] SQUALANE [] METHICONE [] POTASSIUM SORBATE [] CHLORPHENESIN [] [+/- TITANIUM DIOXIDE (CI 77891) [] IRON OXIDES (CI 77491, CI 77492, CI 77499) [] BISMUTH OXYCHLORIDE (CI 77163) [] BLUE 1 (CI 42090) [] CARMINE (CI 75470) [] CHROMIUM HYDROXIDE GREEN (CI 77289) [] CHROMIUM OXIDE GREENS (CI 77288) [] FERRIC FERROCYANIDE (CI 77510) [] YELLOW 5 LAKE (CI 19140) [] ZINC OXIDE (CI 77947)
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MINERALIZE SPF 15 FOUNDATION LOOSE
titanium dioxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:40046-0031 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 7.0 g in 100 g Inactive Ingredients Ingredient Name Strength MICA (UNII: V8A1AW0880) BARLEY BRAN (UNII: 2TXV6Z67KP) WHEAT GERM OIL (UNII: 14C97E680P) ASCORBYL PALMITATE (UNII: QN83US2B0N) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SQUALANE (UNII: GW89575KF9) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) CHLORPHENESIN (UNII: I670DAL4SZ) FERRIC OXIDE RED (UNII: 1K09F3G675) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) COCHINEAL (UNII: TZ8Z31B35M) CHROMIC OXIDE (UNII: X5Z09SU859) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) ALUMINUM OXIDE (UNII: LMI26O6933) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:40046-0031-1 1 in 1 CARTON 1 NDC:40046-0031-2 8.5 g in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 09/01/2007 Labeler - MAKEUP ART COSMETICS (010597206) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 205952385 manufacture Establishment Name Address ID/FEI Business Operations ESTEE LAUDER N.V. 370151326 manufacture Establishment Name Address ID/FEI Business Operations Len-Ron Manufacturing Division of Aramis Inc. 809771152 manufacture Establishment Name Address ID/FEI Business Operations Aramis Inc. 042918826 manufacture Establishment Name Address ID/FEI Business Operations Northtec Bristol 949264774 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Northtec Keystone 618107429 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Estee Lauder Pennsylvania Distribution Center 2 828534516 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics, Ltd. 255175580 manufacture Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics, Ltd 253616536 manufacture Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Distribution Center 208579636 repack, relabel Establishment Name Address ID/FEI Business Operations Estee Lauder Kabushiki Kaisha 712808195 relabel, repack Establishment Name Address ID/FEI Business Operations Whitman Laboratories Ltd. 216866277 manufacture Establishment Name Address ID/FEI Business Operations Aveda Corporation 071352058 manufacture Establishment Name Address ID/FEI Business Operations CRYSTAL CLAIRE COSMETICS 205493484 manufacture