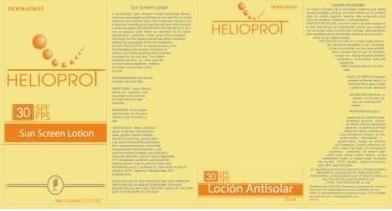
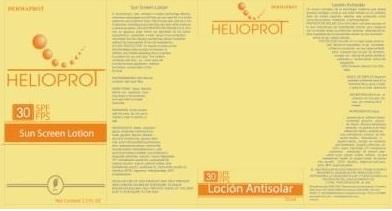
Label: DERMAPROT HELIOPROT AB SPF 30- octinoxate,titanium dioxide,oxybenzone,avobenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 66854-026-01, 66854-026-02, 66854-026-03 - Packager: SPAI-SONS PHARMACEUTICAL INTERNATIONAL COSMETICS
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 7, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DERMAPROT HELIOPROT AB SPF 30
octinoxate,titanium dioxide,oxybenzone,avobenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66854-026 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 mL in 100 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 3 mL in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2.5 mL in 100 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 1.5 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GLYCERIN (UNII: PDC6A3C0OX) ALUMINUM OXIDE (UNII: LMI26O6933) DIMETHICONE (UNII: 92RU3N3Y1O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ALOE VERA LEAF (UNII: ZY81Z83H0X) CHAMOMILE (UNII: FGL3685T2X) CETYL ALCOHOL (UNII: 936JST6JCN) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) OCTYLDODECANOL (UNII: 461N1O614Y) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) POLYSORBATE 20 (UNII: 7T1F30V5YH) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) CHAMOMILE FLOWER OIL (UNII: 60F80Z61A9) EDETATE DISODIUM (UNII: 7FLD91C86K) METHYLPARABEN (UNII: A2I8C7HI9T) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66854-026-01 10 mL in 1 BOTTLE 2 NDC:66854-026-02 45 mL in 1 BOTTLE 3 NDC:66854-026-03 70 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 06/30/2012 Labeler - SPAI-SONS PHARMACEUTICAL INTERNATIONAL COSMETICS (880172184) Registrant - SPAI-SONS PHARMACEUTICAL INTERNATIONAL COSMETICS (880172184) Establishment Name Address ID/FEI Business Operations SPAI-SONS PHARMACEUTICAL INTERNATIONAL COSMETICS 880172184 analysis(66854-026) , manufacture(66854-026) , label(66854-026) , pack(66854-026)