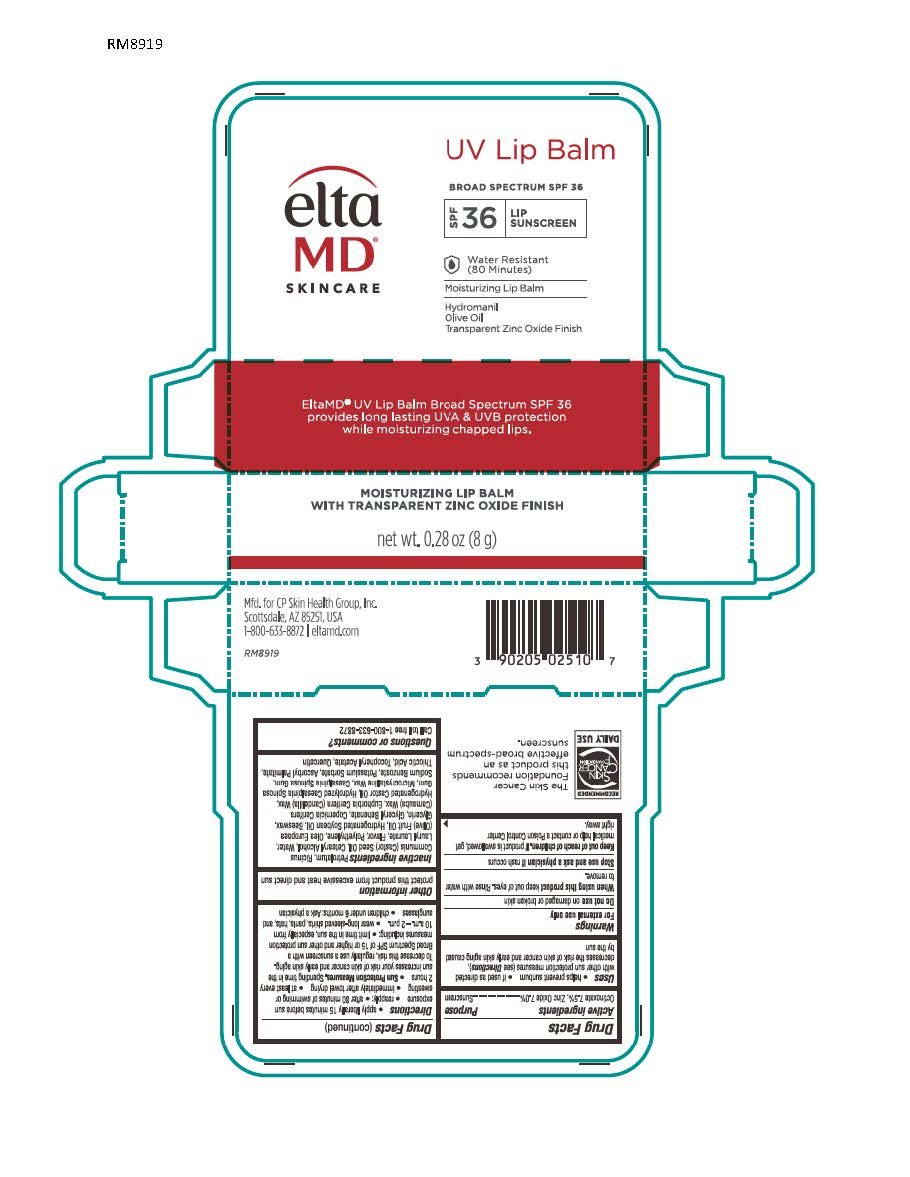
Label: ELTAMD UV LIP BALM SPF31- zinc oxide and octinoxate sunscreen lotion
- NDC Code(s): 72043-2510-8
- Packager: CP Skin Health, Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 25, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Warnings
- Active Ingredients
- Uses
- Uses
-
Directions
apply liberally 15 minutes before sun exposure reapply: after 80 minutes of swimming or sweating immediately after towel drying at lest every 2 hours Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF of 15 or higher and other sun protection measures including: limit time int he sun, especially from 10 a.m. - 2 p.m. wear long-sleeve shirts, pants, hats and sunglasses children under 6 months: Ask a physician
-
Inactive Ingredients
Petrolatum, Ricinus Communis (Castor) Seed Oil, Cetearyl Alcohol, Lauryl Laurate, Purified Water, Flavor, Polyethylene, Hydrogenated Castor Oil, Olea Europaea (Olive) Fruit Oil, Cera Alba, Glycerin, Copernicia Cerifera (Carnauba) Wax, Glyceryl Behenate, Microcrystalline Wax, Quercetin, Thiotic Acid, Ascorbyl Palmitate, Tocopheryl Acetate, Euphorbia Cerifera (Candelilla) Wax, Hydrogenated Soybean Oil, Hydrolyzed Caesalpinia Spinosa Gum, Glyceryl Stearate, Caesalpinia Spinosa Gum
- KEEP OUT OF REACH OF CHILDREN
- Labeling
-
INGREDIENTS AND APPEARANCE
ELTAMD UV LIP BALM SPF31
zinc oxide and octinoxate sunscreen lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72043-2510 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 50 g in 1000 g OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 g in 1000 g Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PETROLATUM (UNII: 4T6H12BN9U) CASTOR OIL (UNII: D5340Y2I9G) LAURYL LAURATE (UNII: GPW77G0937) OLIVE OIL (UNII: 6UYK2W1W1E) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) WHITE WAX (UNII: 7G1J5DA97F) CARNAUBA WAX (UNII: R12CBM0EIZ) CANDELILLA WAX (UNII: WL0328HX19) HYDROGENATED SOYBEAN OIL (UNII: A2M91M918C) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) GLYCERYL DIBEHENATE (UNII: R8WTH25YS2) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) ASCORBYL PALMITATE (UNII: QN83US2B0N) THIOCTIC ACID (UNII: 73Y7P0K73Y) QUERCETIN (UNII: 9IKM0I5T1E) CAESALPINIA SPINOSA RESIN (UNII: WL3883U2PO) GLYCERIN (UNII: PDC6A3C0OX) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72043-2510-8 8 g in 1 TUBE; Type 0: Not a Combination Product 01/10/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/10/2018 Labeler - CP Skin Health, Inc (611921669) Registrant - Swiss-American CDMO, LLC (080170933) Establishment Name Address ID/FEI Business Operations Swiss-American CDMO, LLC 080170933 manufacture(72043-2510)