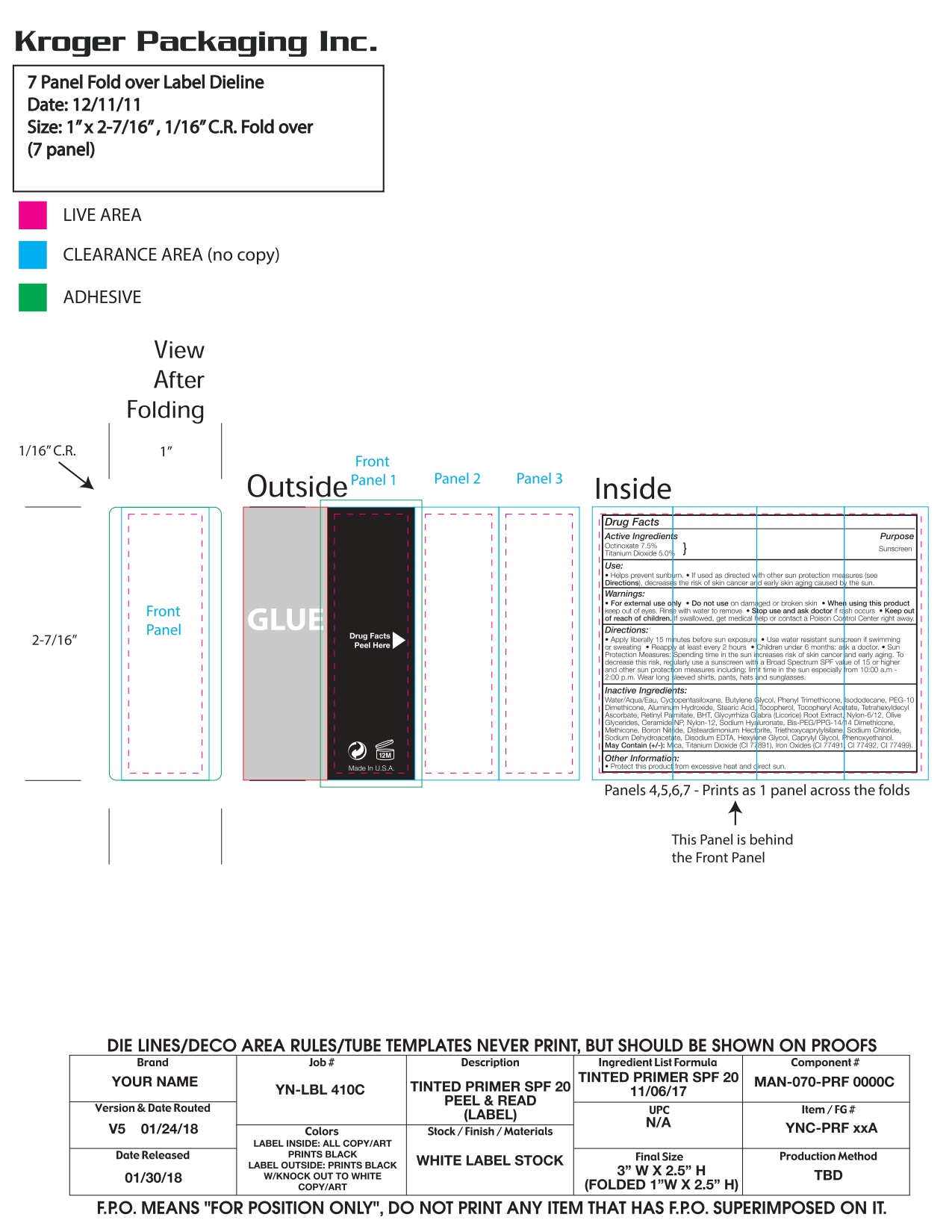
Label: YOUR NAME TINTED PRIMER BROAD SPECTRUM SPF 20- MEDIUM- your name tinted primer cream
- NDC Code(s): 59735-186-01
- Packager: MANA Products, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 5, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Actives
- Purpose
- USE
- WARNINGS
- INDICATIONS & USAGE
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
Apply liberally 15 minutes before sun exposure.
Use water resistant sunscreen if swimming or sweating.Reapply at least every 2 hours.
Children under 6 months: ask a doctor.
Sun Protection Measures: Spending time in the sun increases risk of skin cancer and early aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including: Limit time in the sun especially from 10:00 am - 2:00 pm. Wear long sleeved shirts, pants, hats and sunglasses. -
INACTIVE INGREDIENTS
Water, Cyclopentasiloxane, Butylene Glycol, Titanium Dioxide, Zinc Oxide, Octyldodecyl Neopentanoate, PEG/PPG-18/18 Dimethicone, Dimethicone, Tocopherol, Tetrahexyldecyl Ascorbate, Retinyl Palmitate, Camellia Sinensis Leaf Extract, Chamomilla Recutita (Matricaria) Flower Extract, Ginkgo Biloba Leaf Extract, Panax Ginseng Root Extract, Octyldodecanol, Boron Nitride, Dextrin Palmitate, Palmitic Acid, Methicone, Stearic Acid, Magnesium Chloride, Potassium Chloride, Sodium Chloride, Zinc Chloride, Magnesium Ascorbyl Phosphate, Sodium Hyaluronate, Phytantriol, Glycerin, Triethoxycaprylylsilane, Lysine, Aluminum Hydroxide, Disodium EDTA, Hexylene Glycol, Potassium Sorbate, Sodium Dehydroacetate, Caprylyl Glycol, Phenoxyethanol
MAY CONTAIN (+/-):
Mica, Titanium Dioxide (CI 77891), Iron Oxides (CI 77491, CI 77492, CI 77499)
- OTHER INFORMATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
YOUR NAME TINTED PRIMER BROAD SPECTRUM SPF 20- MEDIUM
your name tinted primer creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59735-186 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 36 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 36 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) ISODODECANE (UNII: A8289P68Y2) TOCOPHEROL (UNII: R0ZB2556P8) CERAMIDE NP (UNII: 4370DF050B) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HEXYLENE GLYCOL (UNII: KEH0A3F75J) SODIUM DEHYDROACETATE (UNII: 8W46YN971G) EDETATE DISODIUM (UNII: 7FLD91C86K) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE RED (UNII: 1K09F3G675) LICORICE (UNII: 61ZBX54883) HYALURONIC ACID (NON-ANIMAL STABILIZED) (UNII: B7SG5YV2SI) NYLON-12 (UNII: 446U8J075B) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PEG-10 DIMETHICONE (200 CST) (UNII: 287GF3Y3WC) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) SODIUM CHLORIDE (UNII: 451W47IQ8X) STEARIC ACID (UNII: 4ELV7Z65AP) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) BORON NITRIDE (UNII: 2U4T60A6YD) METHICONE (20 CST) (UNII: 6777U11MKT) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) OLIVE OIL (UNII: 6UYK2W1W1E) PHENOXYETHANOL (UNII: HIE492ZZ3T) BIS-PEG/PPG-14/14 DIMETHICONE (UNII: X2I70H0QJE) MICA (UNII: V8A1AW0880) NYLON 612 (MW 14000) (UNII: E7LN56Z3RX) Product Characteristics Color brown Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59735-186-01 1 in 1 CARTON 02/01/2011 1 36 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 02/01/2011 Labeler - MANA Products, Inc. (078870292) Establishment Name Address ID/FEI Business Operations MANA Products, Inc. 078870292 manufacture(59735-186) Establishment Name Address ID/FEI Business Operations MANA Products,Inc. 032870270 manufacture(59735-186)