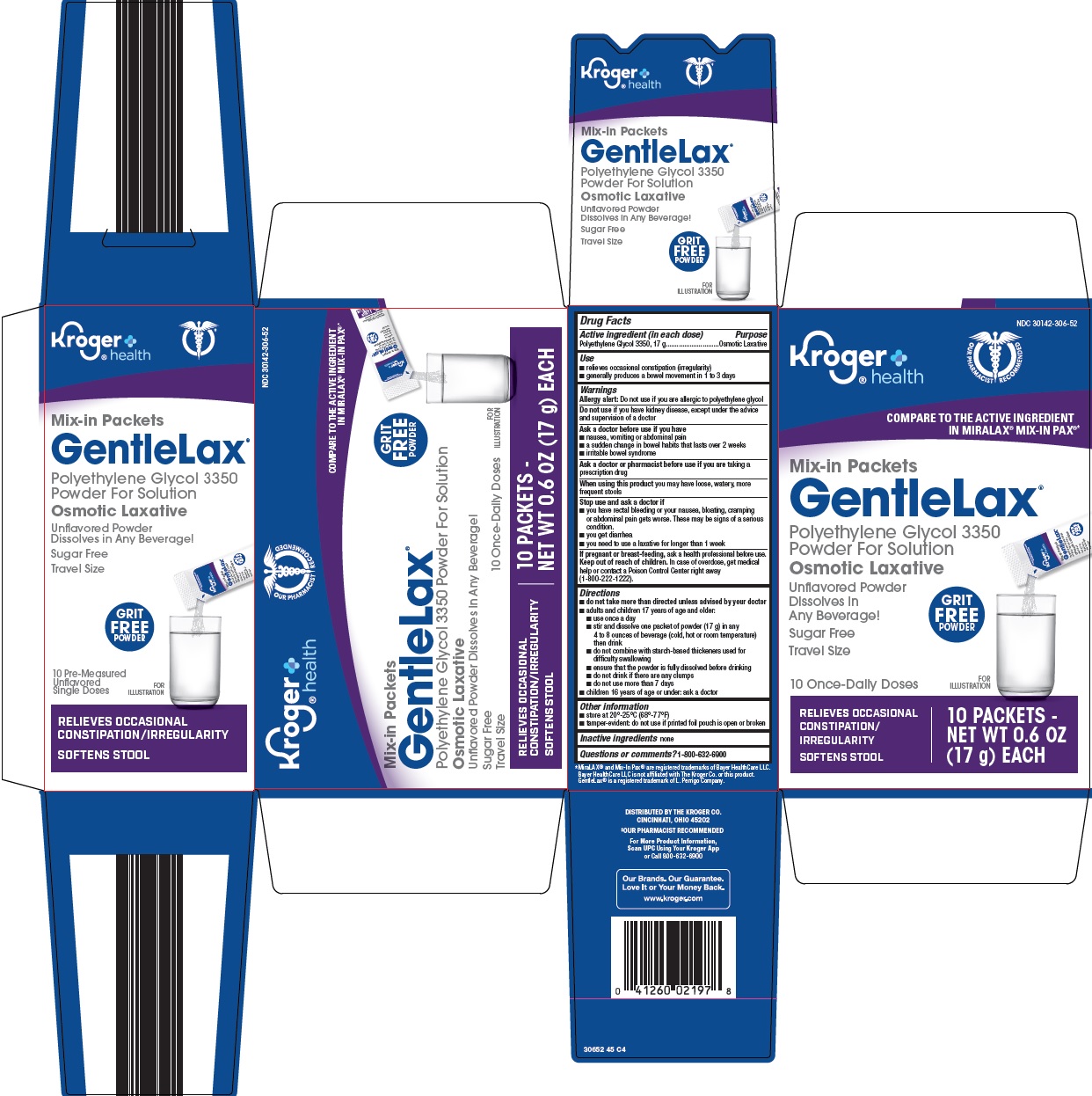
Label: GENTLE LAX- polyethylene glycol 3350 powder, for solution
-
NDC Code(s):
30142-306-01,
30142-306-02,
30142-306-03,
30142-306-04, view more30142-306-09, 30142-306-17, 30142-306-19, 30142-306-52
- Packager: Kroger Company
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated May 21, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each dose)
- Purpose
- Use
-
Warnings
Allergy alert: Do not use if you are allergic to polyethylene glycol
Ask a doctor before use if you have
- •
- nausea, vomiting or abdominal pain
- •
- a sudden change in bowel habits that lasts over 2 weeks
- •
- irritable bowel syndrome
-
Directions
- •
- do not take more than directed unless advised by your doctor
- •
- adults and children 17 years of age and older:
- •
- use once a day
- •
- stir and dissolve one packet of powder (17 g) in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- •
- do not combine with starch-based thickeners used for difficulty swallowing
- •
- ensure that the powder is fully dissolved before drinking
- •
- do not drink if there are any clumps
- •
- do not use more than 7 days
- •
- children 16 years of age or under: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
Kroger® health
Mix-in Packets
GentleLax®
Polyethylene Glycol 3350
Powder For Solution
Osmotic Laxative
Unflavored Powder
Dissolves in Any Beverage!
Sugar Free
Travel Size
GRIT FREE POWDER
10 Pre-Measured Unflavored Single Doses
10 Once-Daily Doses
RELIEVES OCCASIONAL CONSTIPATION/IRREGULARITY
SOFTENS STOOL
10 PACKETS – NET WT 0.6 OZ (17 g) EACH
-
INGREDIENTS AND APPEARANCE
GENTLE LAX
polyethylene glycol 3350 powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:30142-306 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 17 g in 17 g Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:30142-306-03 510 g in 1 BOTTLE; Type 0: Not a Combination Product 10/07/2009 2 NDC:30142-306-01 119 g in 1 BOTTLE; Type 0: Not a Combination Product 10/07/2009 3 NDC:30142-306-02 238 g in 1 BOTTLE; Type 0: Not a Combination Product 10/07/2009 4 NDC:30142-306-17 289 g in 1 BOTTLE; Type 0: Not a Combination Product 11/16/2014 11/16/2014 5 NDC:30142-306-19 595 g in 1 BOTTLE; Type 0: Not a Combination Product 11/18/2013 02/28/2022 6 NDC:30142-306-04 850 g in 1 BOTTLE; Type 0: Not a Combination Product 08/13/2013 09/25/2016 7 NDC:30142-306-09 765 g in 1 BOTTLE; Type 0: Not a Combination Product 08/14/2015 8 NDC:30142-306-52 10 in 1 CARTON 05/21/2024 8 17 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090685 10/07/2009 Labeler - Kroger Company (006999528)