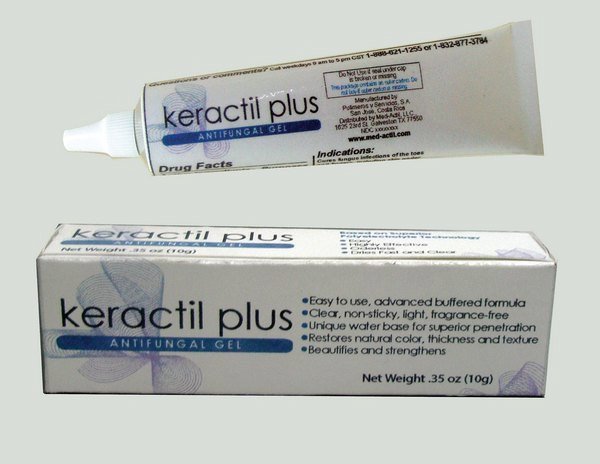
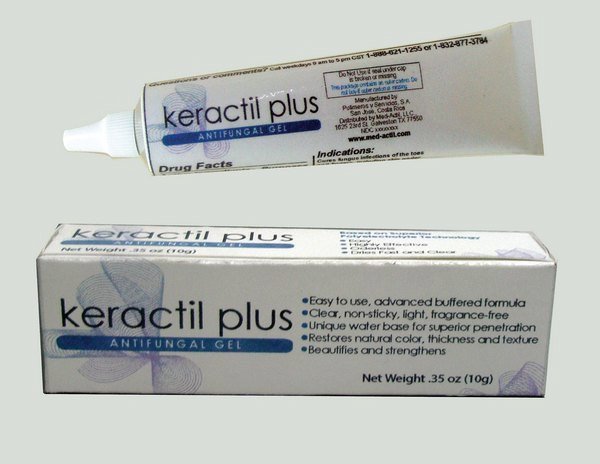
Label: KERACTIL PLUS- antifungal gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 69492-014-06 - Packager: POLIMEROS Y SERVICIOS S.A.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 5, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Representative sample image of carton/container label
General view of box content of labeling
Caja con cambios Keractil Plus.jpg
Representative sample image of carton/container label
Keractil Fondo gris.jpg
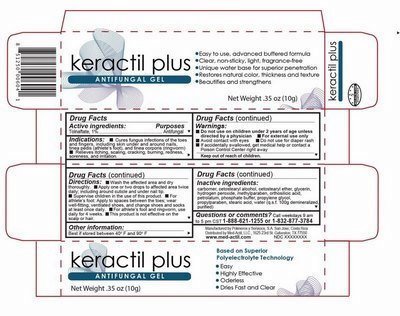
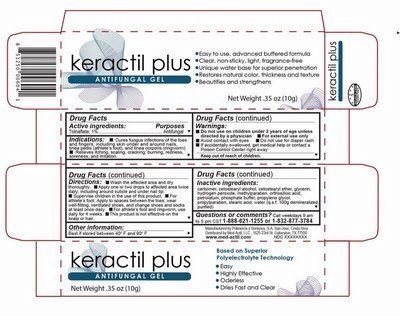
Detail showing Active Ingredient from package
Information shown both on container label and package
Active Ingredients- Keractil plus.jpg

Detail showing Inctive Ingredient from package
Infiomaion shown both on content of label and package
Inactive ingredients.jpg

Detail showing Purpose within Active Ingredients from package
Information shown both on container label and package
Purpose.jpg

Detail showing Instructions for use and named Uses from package
Ibformation shown both on container label and package
Directions.jpg

Detail showing Dosage and Administration within Directions from package
Information shown both on container label and package
Dosage and administration.jpg

-
INGREDIENTS AND APPEARANCE
KERACTIL PLUS
antifungal gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69492-014 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) HYDROGEN PEROXIDE (UNII: BBX060AN9V) METHYLPARABEN (UNII: A2I8C7HI9T) ORTHOSILICIC ACID (UNII: 623B93YABH) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69492-014-06 1 in 1 BOX 03/04/2019 1 10 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 03/09/2016 Labeler - POLIMEROS Y SERVICIOS S.A. (815740251) Establishment Name Address ID/FEI Business Operations POLIMEROS Y SERVICIOS S.A. 815740251 manufacture(69492-014)