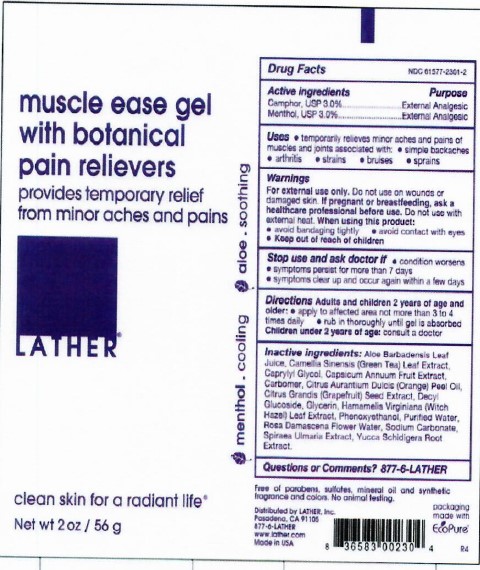
Label: MUSCLE EASE- menthol camphor gel
- NDC Code(s): 61577-2301-2, 61577-2301-4
- Packager: SOMBRA COSMETICS INC.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 14, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Keep out of reach of children
- Uses
- Warnings
- Directions
-
Inactive Ingredients
Aloe Barbadensis leaf Juice, Camellia Sinensis (Green Tea) Leaf Extract, Caprylyl Glycol, Capsicum Annuum Fruit Extract, Carbomer, Citrus Aurantium Dulcis (Orange) Peel Oil, Citrus Grandis (Grapefruit) Seed Extract, Decyl Glucoside, Filipendula Ulmaria (Queen of the Praire) Flower Extract, Glycerin, Hamamelis Virginiana (Witch Hazel) Leaf Extract, Phenoxyethanol, Purified Water, Rosa Damascena Flower Water, Sodium Carbonate, Yucca Schidigera Stem Extract.
- Questions or Comments
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MUSCLE EASE
menthol camphor gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61577-2301 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.03 g in 1 g CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 0.03 g in 1 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPSICUM (UNII: 00UK7646FG) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) WATER (UNII: 059QF0KO0R) GRAPE SEED OIL (UNII: 930MLC8XGG) GREEN TEA LEAF (UNII: W2ZU1RY8B0) ORANGE PEEL (UNII: TI9T76XD44) FILIPENDULA ULMARIA FLOWER (UNII: 06L18L32G6) ROSA DAMASCENA FLOWER (UNII: JWB78P295A) GLYCERIN (UNII: PDC6A3C0OX) WITCH HAZEL (UNII: 101I4J0U34) CAPRYLYL GLYCOL (UNII: 00YIU5438U) YUCCA SCHIDIGERA ROOT (UNII: E2H9ET15AT) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM CARBONATE (UNII: 45P3261C7T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61577-2301-4 113.4 g in 1 JAR; Type 0: Not a Combination Product 02/14/2023 2 NDC:61577-2301-2 56.7 g in 1 TUBE; Type 0: Not a Combination Product 02/14/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/14/2023 Labeler - SOMBRA COSMETICS INC. (097464309) Establishment Name Address ID/FEI Business Operations SOMBRA COSMETICS INC. 097464309 manufacture(61577-2301) , label(61577-2301)