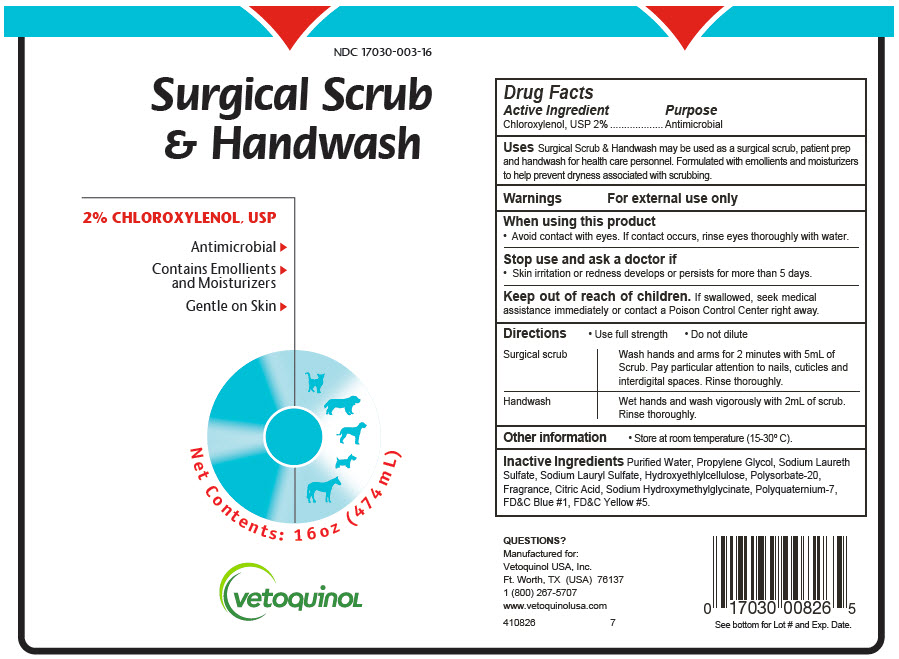
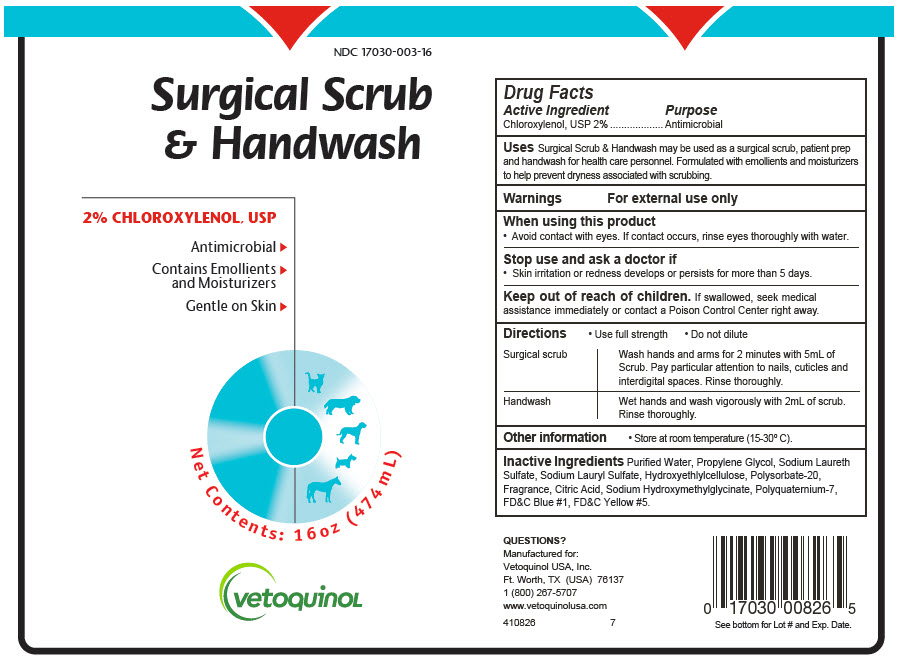
Label: SURGICAL SCRUB AND HANDWASH- chloroxylenol soap
- NDC Code(s): 17030-003-10, 17030-003-16
- Packager: Vetoquinol USA, Inc.
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 9, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- INDICATIONS & USAGE
- Warnings
- Directions
- Other information
- SPL UNCLASSIFIED SECTION
- QUESTIONS?
- PRINCIPAL DISPLAY PANEL - 474 mL Bottle Label
-
INGREDIENTS AND APPEARANCE
SURGICAL SCRUB AND HANDWASH
chloroxylenol soapProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:17030-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Chloroxylenol (UNII: 0F32U78V2Q) (Chloroxylenol - UNII:0F32U78V2Q) Chloroxylenol 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURETH-3 SULFATE (UNII: BPV390UAP0) SODIUM LAURYL SULFATE (UNII: 368GB5141J) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) POLYSORBATE 20 (UNII: 7T1F30V5YH) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM HYDROXYMETHYLGLYCINATE (UNII: DIG6BWZ9XT) POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600000 MW) (UNII: 0L414VCS5Y) Product Characteristics Color BLUE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17030-003-16 474 mL in 1 BOTTLE, PLASTIC 2 NDC:17030-003-10 3790 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 03/10/2017 Labeler - Vetoquinol USA, Inc. (106824209) Registrant - Vetoquinol USA, Inc. (106824209) Establishment Name Address ID/FEI Business Operations N.S.Chemicals 650469732 API MANUFACTURE, ANALYSIS Establishment Name Address ID/FEI Business Operations Tropichem Research Labs, LLC dba Vetio 603123068 MANUFACTURE, ANALYSIS, LABEL, PACK