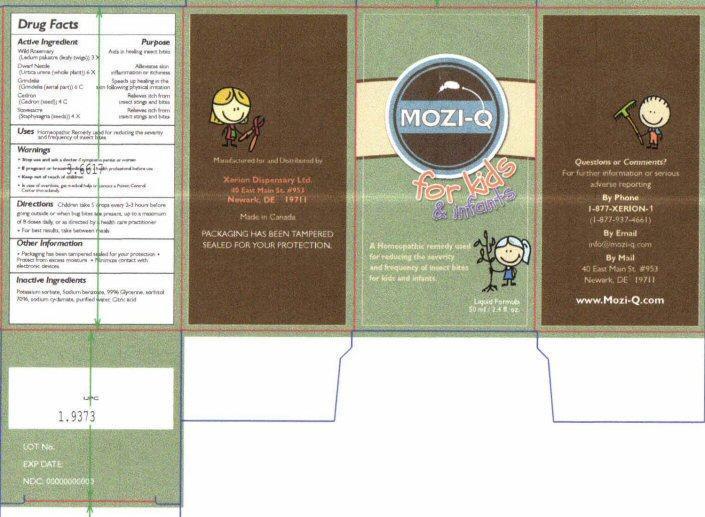
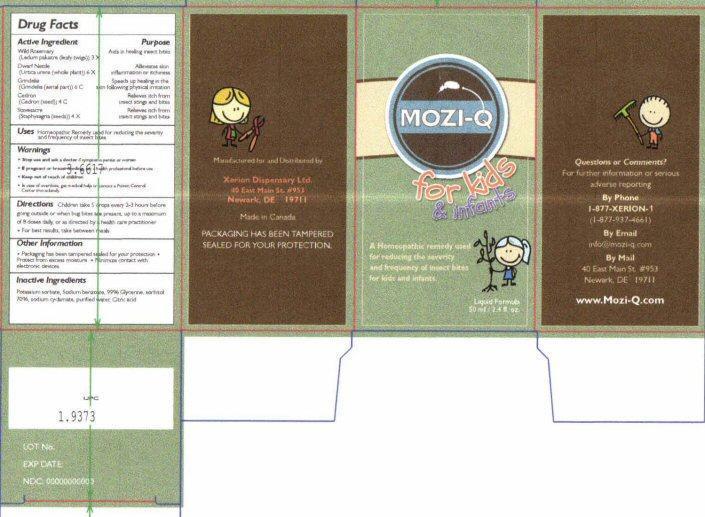
Label: MOZI-QFOR KIDS AND INFA FOR KIDS AND INFANTS- ledum palustre twig, urtica urens, simaba cedron seed, delphinium staphisagria seed, grindelia hirsutula flowering top liquid
- NDC Code(s): 53590-102-24
- Packager: Xerion Dispensary Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts Active Ingredients
- Purpose
- Warnings Keep out of reach of children
- Uses
- Warnings
- Directions
- Other Information
- Inactive Ingredients
-
Product Label
MOZI-Q for kids & infants
MOZI-Q for kids and infants
A Homeopathic remedy used for reducing the severity and frequency of insect bites for kids and infants
Liquid Formula 50 ml / 2.4 fl. oz.
Questions or Comments?
For further information or serious adverse reporting
By Phone 1-877-XERION-1 (1-877-937-4661)
By Email info@mozi-q.com
By Mail 40 East Main St # 953 Newark, DE 19711
www.Mozi-Q.com
Manufactured for and Distributed by
Xerion Dispensary Ltd.
40 East Main St #953
Newark, DE 19711
Made in Canada
PACKAGING HAS BEEN TAMPERED SEALED FOR YOUR PROTECTION
LOT No.
EXP DATE:
NDC 00000000000
-
INGREDIENTS AND APPEARANCE
MOZI-QFOR KIDS AND INFA FOR KIDS AND INFANTS
ledum palustre twig, urtica urens, simaba cedron seed, delphinium staphisagria seed, grindelia hirsutula flowering top liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53590-102 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEDUM PALUSTRE TWIG (UNII: 877L01IZ0P) (LEDUM PALUSTRE TWIG - UNII:877L01IZ0P) LEDUM PALUSTRE TWIG 3 [hp_X] in 1 mL URTICA URENS (UNII: IHN2NQ5OF9) (URTICA URENS - UNII:IHN2NQ5OF9) URTICA URENS 6 [hp_X] in 1 mL GRINDELIA HIRSUTULA FLOWERING TOP (UNII: IDB0NAZ6AI) (GRINDELIA HIRSUTULA FLOWERING TOP - UNII:IDB0NAZ6AI) GRINDELIA HIRSUTULA FLOWERING TOP 6 [hp_C] in 1 mL SIMABA CEDRON SEED (UNII: KHI40967MV) (SIMABA CEDRON SEED - UNII:KHI40967MV) SIMABA CEDRON SEED 4 [hp_C] in 1 mL DELPHINIUM STAPHISAGRIA SEED (UNII: 00543AP1JV) (DELPHINIUM STAPHISAGRIA SEED - UNII:00543AP1JV) DELPHINIUM STAPHISAGRIA SEED 4 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM BENZOATE (UNII: OJ245FE5EU) GLYCERIN (UNII: PDC6A3C0OX) SORBITOL (UNII: 506T60A25R) SODIUM CYCLAMATE (UNII: 1I6F42RME1) WATER (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53590-102-24 50 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 02/01/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 02/01/2014 Labeler - Xerion Dispensary Ltd. (250925265) Registrant - Xerion Dispensary Ltd. (250925265)