Label: QUIK-CARE- alcohol solution
-
NDC Code(s):
47593-500-38,
47593-500-41,
47593-500-55,
47593-500-56, view more47593-500-57, 47593-500-59, 47593-500-85
- Packager: Ecolab Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
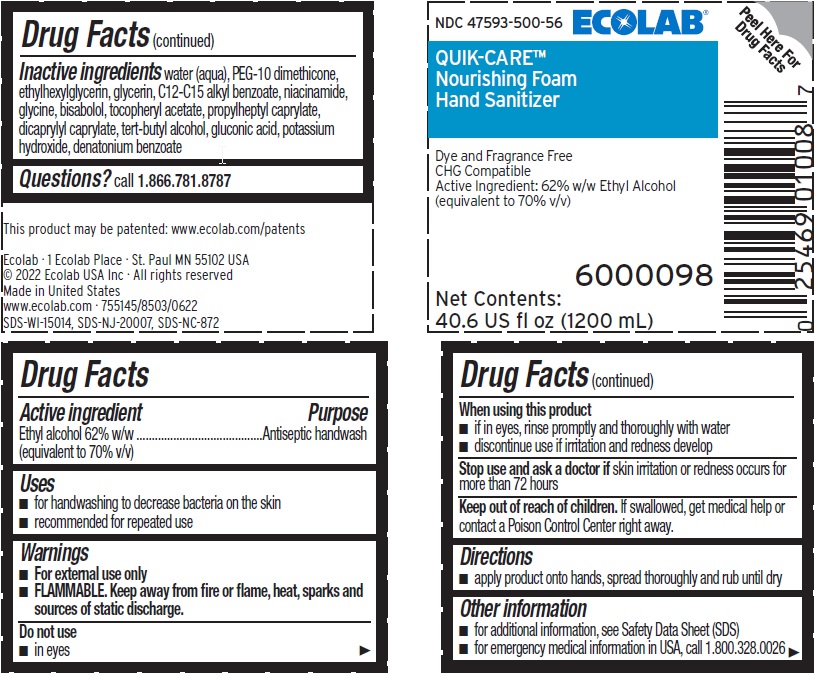
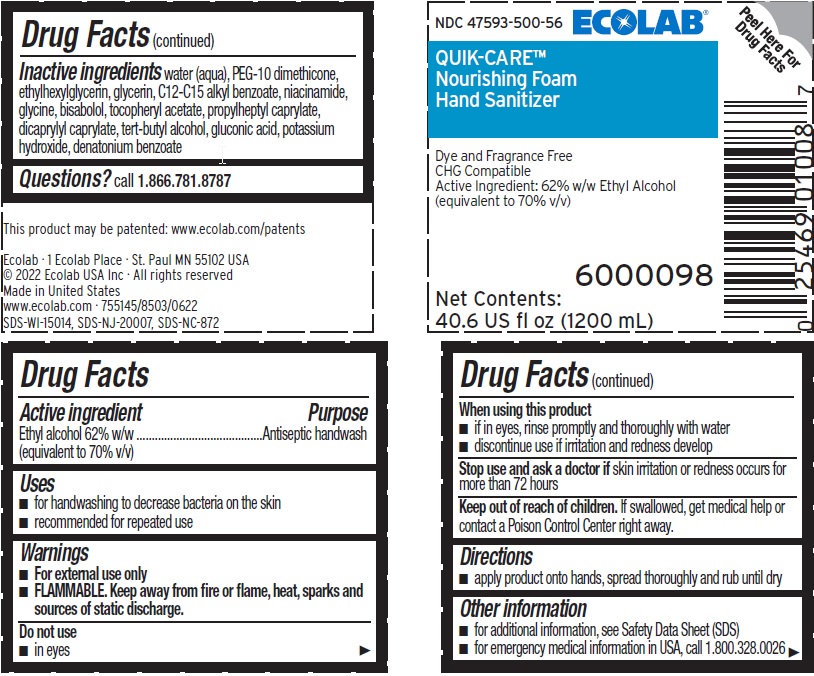
- Active ingredient
- Purpose
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- INACTIVE INGREDIENT
- QUESTIONS
-
Principal Display Panel and Representative Label
ECOLAB®
NDC 47593-500-56
QUIK-CARE®
Nourishing Foam
Hand Sanitizer
Dye and Fragrance Free
CHG Compatible
Active Ingredient: 62% Ethyl Alcohol
(equivalent to 70% v/v)
6000098
Net Contents: 40.6 US fl oz (1200 mL)
This product may be patented: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA
© 2022 Ecolab USA Inc · All rights reserved
Made in United States
www.ecolab.com · 755145/8503/0622
SDS-WI-15014, SDS-NJ-20007, SDS-NC-872
-
INGREDIENTS AND APPEARANCE
QUIK-CARE
alcohol solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:47593-500 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 539.4 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) NIACINAMIDE (UNII: 25X51I8RD4) GLYCINE (UNII: TE7660XO1C) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PROPYLHEPTYL CAPRYLATE (UNII: 991Z19V2OD) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) TERT-BUTYL ALCOHOL (UNII: MD83SFE959) GLUCONIC ACID (UNII: R4R8J0Q44B) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) DENATONIUM BENZOATE (UNII: 4YK5Z54AT2) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:47593-500-55 1000 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 2 NDC:47593-500-57 535 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 3 NDC:47593-500-38 500 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 02/19/2024 4 NDC:47593-500-85 45 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 5 NDC:47593-500-41 750 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 6 NDC:47593-500-59 1200 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 05/03/2018 7 NDC:47593-500-56 1200 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/01/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 02/01/2013 Labeler - Ecolab Inc. (006154611)