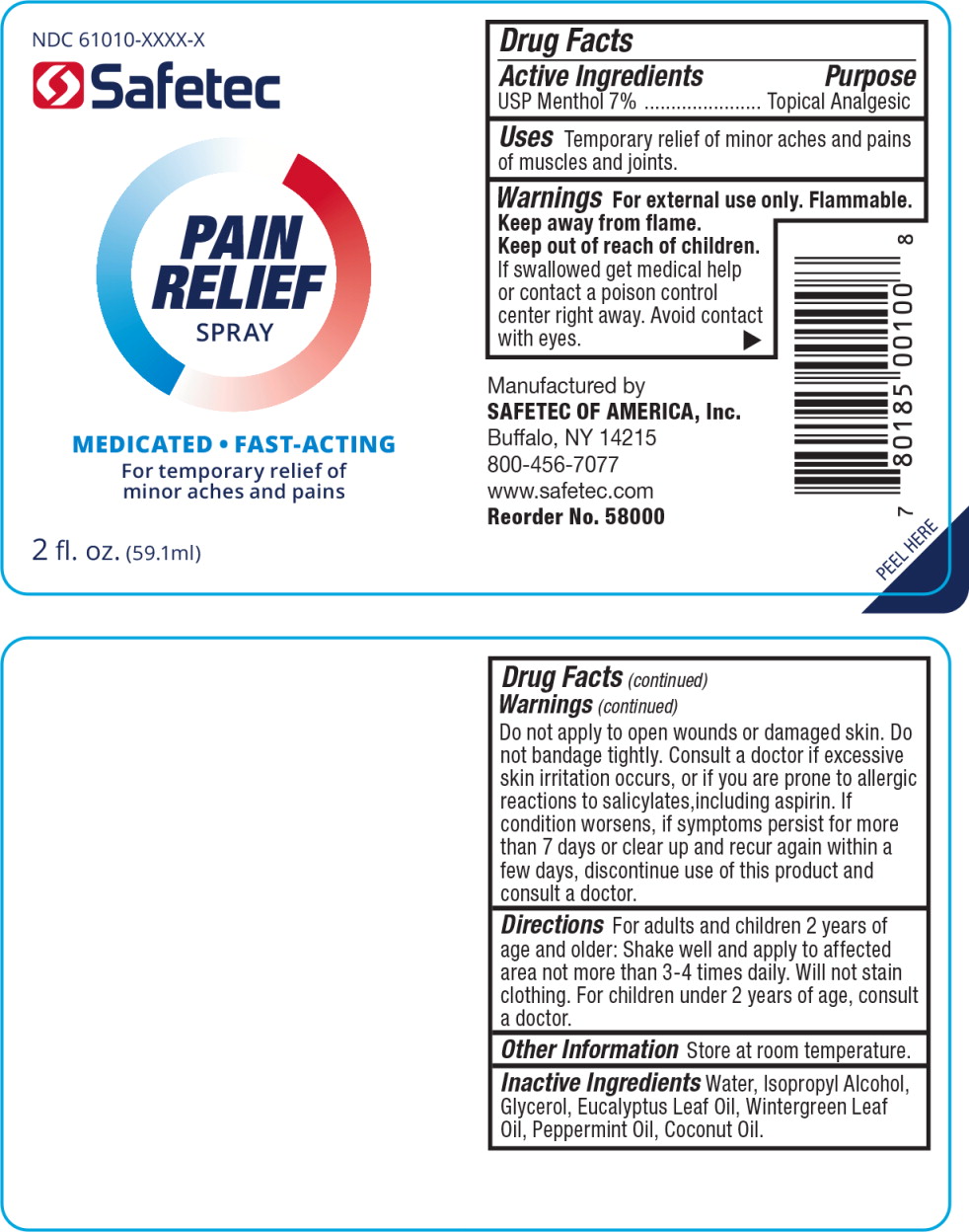
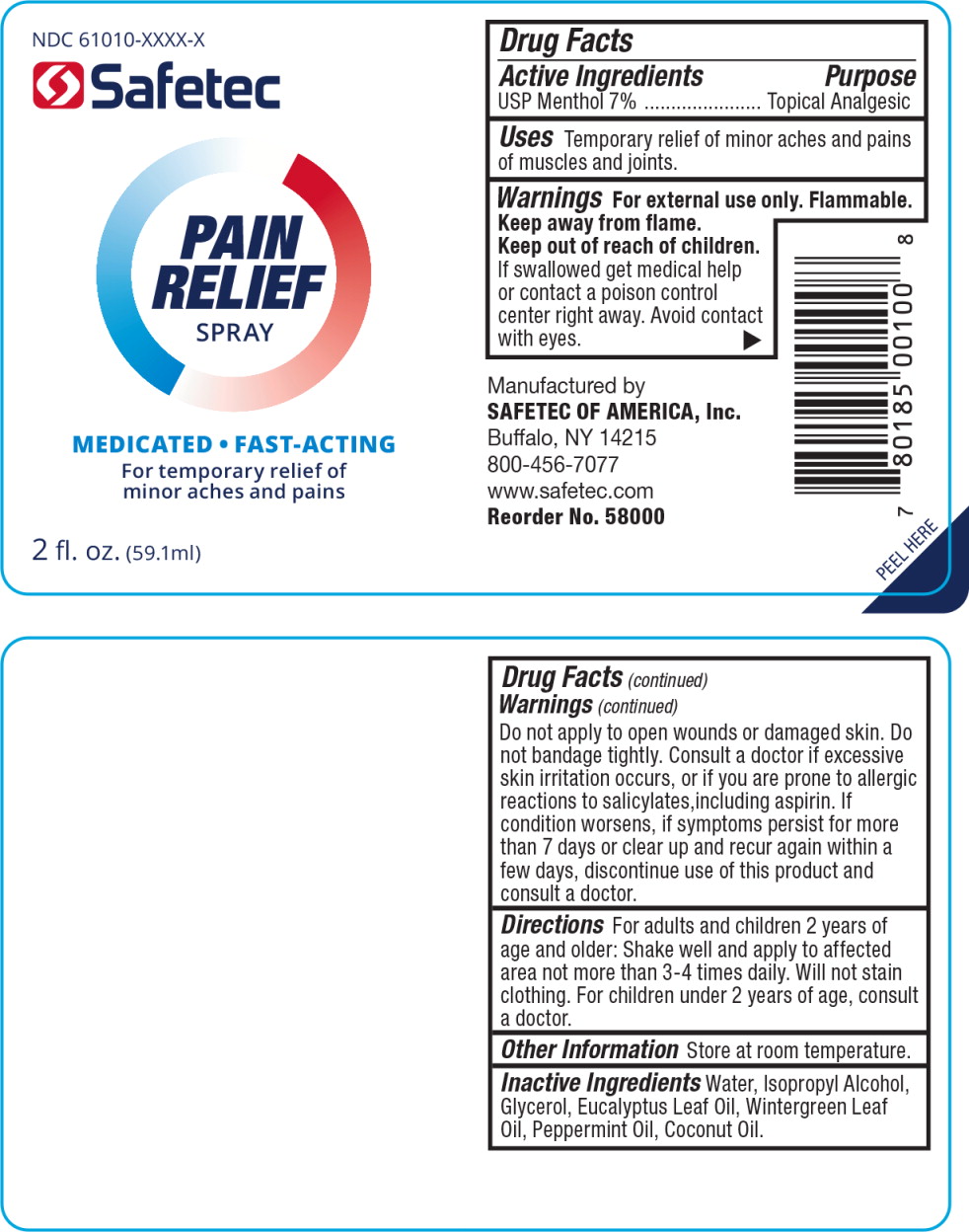
Label: ANALGESIC- menthol spray
- NDC Code(s): 61010-8201-0
- Packager: Safetec of America, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 4, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Directions
- Other Information
- Inactive Ingredients
- Principal Display Panel - Pain Relief Spray Bottle Label
-
INGREDIENTS AND APPEARANCE
ANALGESIC
menthol sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61010-8201 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 70 mg in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ISOPROPYL ALCOHOL (UNII: ND2M416302) GLYCERIN (UNII: PDC6A3C0OX) EUCALYPTUS OIL (UNII: 2R04ONI662) METHYL SALICYLATE (UNII: LAV5U5022Y) PEPPERMINT OIL (UNII: AV092KU4JH) COCONUT OIL (UNII: Q9L0O73W7L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61010-8201-0 0.059 L in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 09/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 09/01/2018 Labeler - Safetec of America, Inc. (874965262) Establishment Name Address ID/FEI Business Operations Safetec of America, Inc 874965262 manufacture(61010-8201)