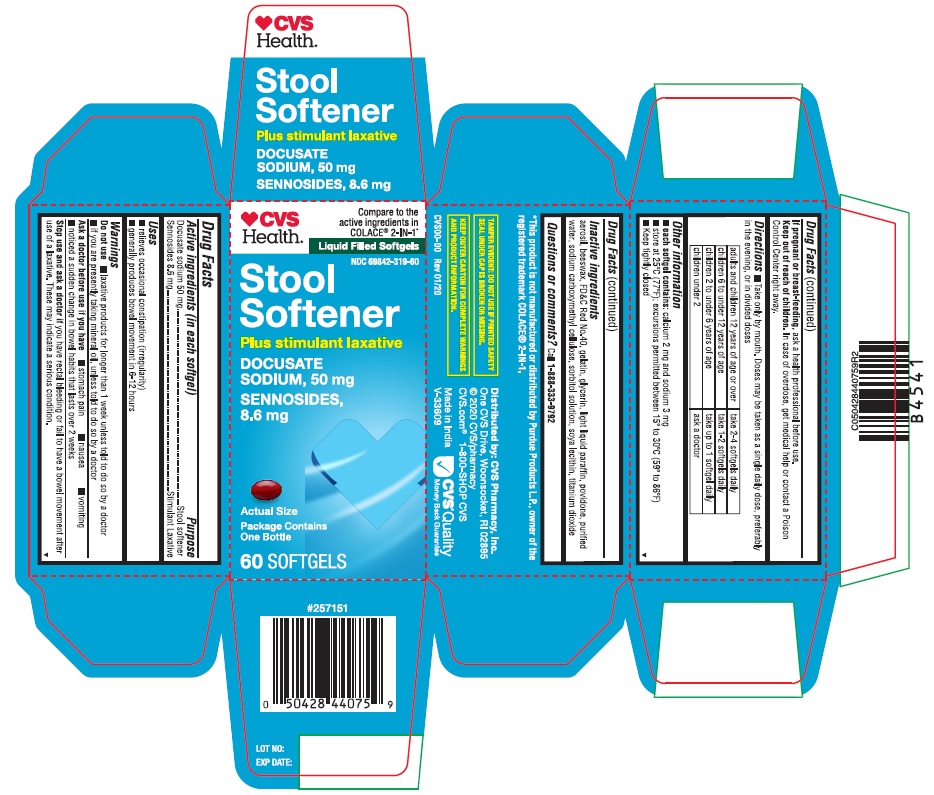
Label: STOOL SOFTENER- docusate sodium and sennosides capsule, gelatin coated
- NDC Code(s): 69842-319-60
- Packager: CVS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 6, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- Uses
-
WARNINGS
Warnings
Do not use
- laxative products for longer than 1 week unless told to do so by a doctor
- if you are presently taking mineral oil, unless told to do so by a doctor
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
-
Directions
- Take only by mouth. Doses may be taken as a single daily dose, preferably in the evening, or in divided doses
adults and children 12 years of age or over take 2-4 softgels daily children 6 to under 12 years of age take 1-2 softgels daily children 2 to under 6 years of age take upto 1 softgel daily children under 2 ask a doctor - Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
♥CVSHealth™
Compare to the
active ingredients in
COLACE® 2-IN-1*Liquid Filled Softgels
NDC 69842-319-60
Stool Softener
DOCUSATE SODIUM, 50 mg
SENNOSIDES, 8.6 mg
Plus stimulant laxativePackage Contains One Bottle
60 SOFTGELS
Actual Size
*This product is not manufactured or distributed by Purdue Products L.P., owner of the
registered trademark COLACE® 2-IN-1.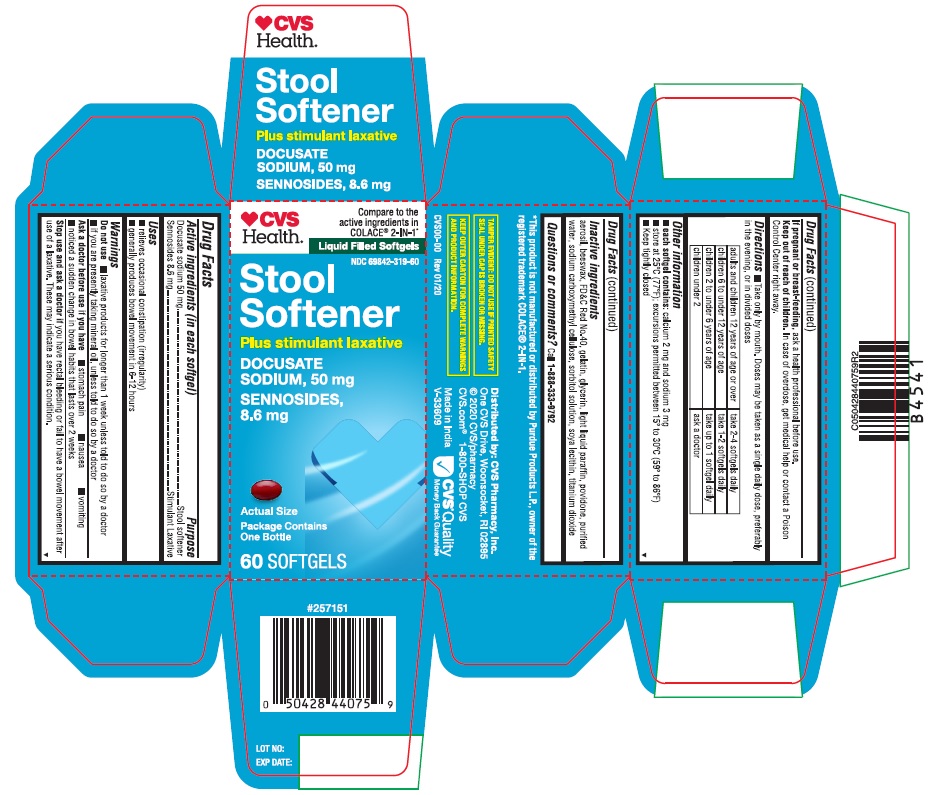
-
INGREDIENTS AND APPEARANCE
STOOL SOFTENER
docusate sodium and sennosides capsule, gelatin coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-319 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) YELLOW WAX (UNII: 2ZA36H0S2V) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) LIGHT MINERAL OIL (UNII: N6K5787QVP) POVIDONE K30 (UNII: U725QWY32X) WATER (UNII: 059QF0KO0R) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) SORBITOL (UNII: 506T60A25R) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color red (OPAQUE) Score no score Shape CAPSULE (SOFTGEL) Size 12mm Flavor Imprint Code 584 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-319-60 1 in 1 CARTON 03/15/2018 1 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 03/15/2018 Labeler - CVS (062312574)