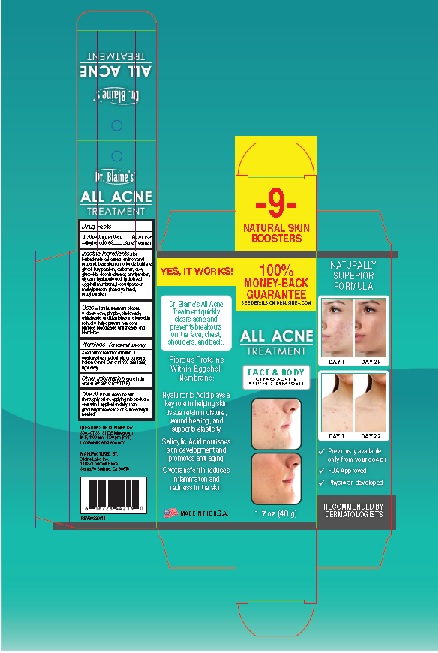
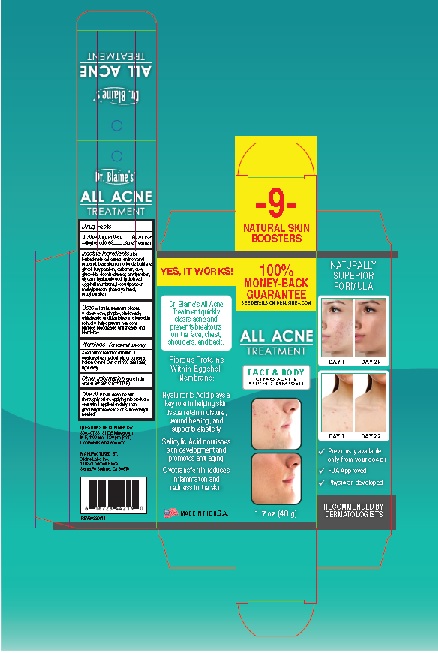
Label: ALL ACNE TREATMENT- salicylic acid gel
- NDC Code(s): 63347-130-01
- Packager: Blaine Labs Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
- QUESTIONS
- INACTIVE INGREDIENT
- Product Label
-
INGREDIENTS AND APPEARANCE
ALL ACNE TREATMENT
salicylic acid gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63347-130 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.5 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) BUTYLPARABEN (UNII: 3QPI1U3FV8) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) WATER (UNII: 059QF0KO0R) ETHYLPARABEN (UNII: 14255EXE39) GLYCERIN (UNII: PDC6A3C0OX) HYALURONIC ACID (UNII: S270N0TRQY) EGG SHELL MEMBRANE (UNII: N7QBR4212V) ISOBUTYLPARABEN (UNII: 0QQJ25X58G) METHYLPARABEN (UNII: A2I8C7HI9T) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63347-130-01 1 in 1 BOX 05/17/2022 1 48 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 05/17/2022 Labeler - Blaine Labs Inc. (017314571) Establishment Name Address ID/FEI Business Operations Blaine Labs Inc. 017314571 manufacture(63347-130)